Redox Chemistry Of Early Earth And The Origin Of Life
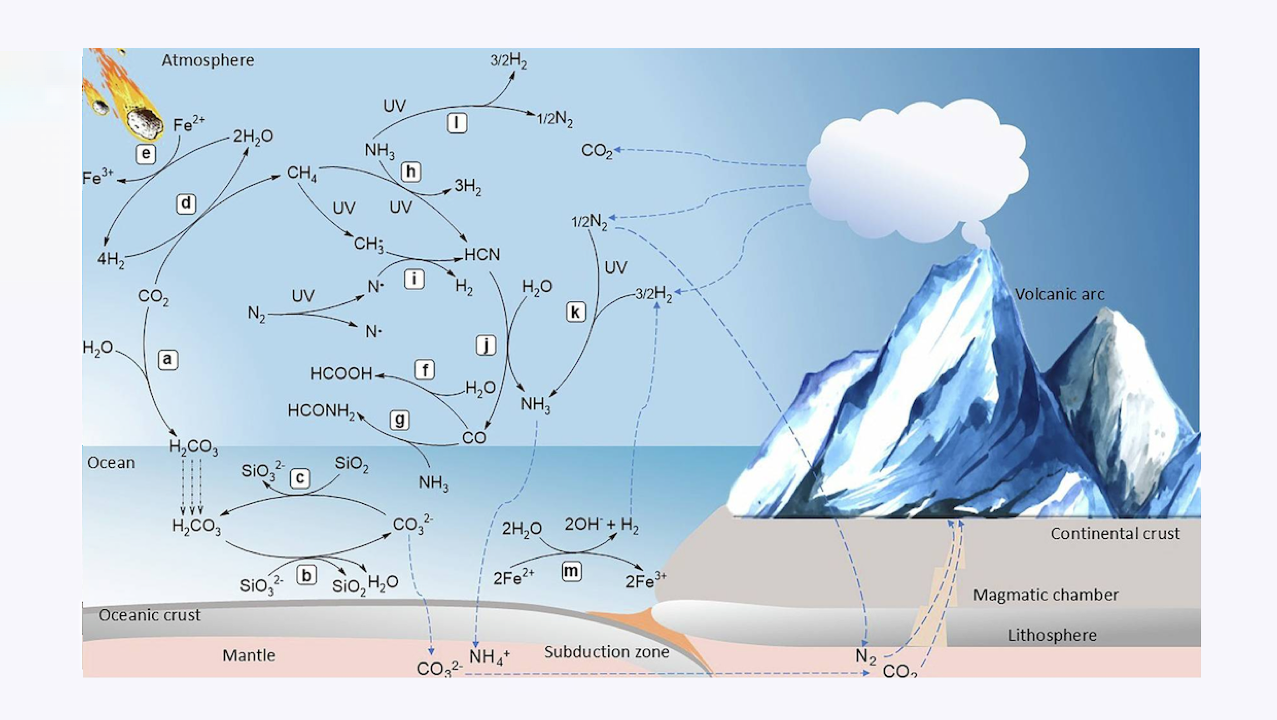
Multiple controversial scenarios have been proposed explaining the emergence of life, both from environmental and chemical perspectives.
These scenarios discuss environmental settings including deep-ocean hydrothermal vents, volcanic lakes, hot springs, and geysers, which could have served as the cradle for life. Environmental conditions varied by energy, chemical elements, minerals, transition metals, dynamic light-dark, hot-cold, and wet-dry cycles for the first organic synthesis.
Other controversies concern the type of organisms that could have appeared first: either heterotrophic or autotrophic. The heterotrophic theory suggests that the first organic compounds were abiotically synthesized from simple inorganic molecules accumulating in the oceans and concentrating in coacervates, which functioned as early pre-cellular structures.
The chemoautotrophic theory argues that life originated from inorganic compounds, rather than from organic matter establishing proto-metabolic pathways in primary producers. All of the above-mentioned theories rely on redox reactions that created necessary conditions and molecules for the origin of life.
This review explores evidences that reconcile the dominant theories, including that: (i) the Hadean atmosphere was weakly oxidizing, but transiently reducing, (ii) hydrothermal systems provided energy for organic synthesis supporting both heterotrophs and chemoautotrophs, (iii) organic compounds were transported between different environments, and (iv) life emerged in multiple local environments.
- Redox chemistry of early Earth and the origin of life, Nature via PubMed (open access)
- Redox chemistry of early Earth and the origin of life, Nature (open access)
Astrobiology, Biochemistry,