Stability and Reactivity of Alternative Nucleobases in Concentrated Sulfuric Acid.

Recent findings demonstrate that concentrated sulfuric acid supports rich organic chemistry, including the stability of the canonical DNA bases adenine, thymine, guanine and cytosine.
Yet, due to full protonation in concentrated sulfuric acid, these bases may not pair as effectively as they do in water. We are therefore motivated to study nucleic acid bases that pair via hydrophobic and van der Waals interactions instead of canonical hydrogen bonding.
Here, we investigate the stability of 14 selected, commercially available alternative nucleobases in concentrated sulfuric acid to evaluate their potential for forming DNA-like polymers in this solvent. The reactivity of compounds 1–14 have not been previously investigated in concentrated sulfuric acid.
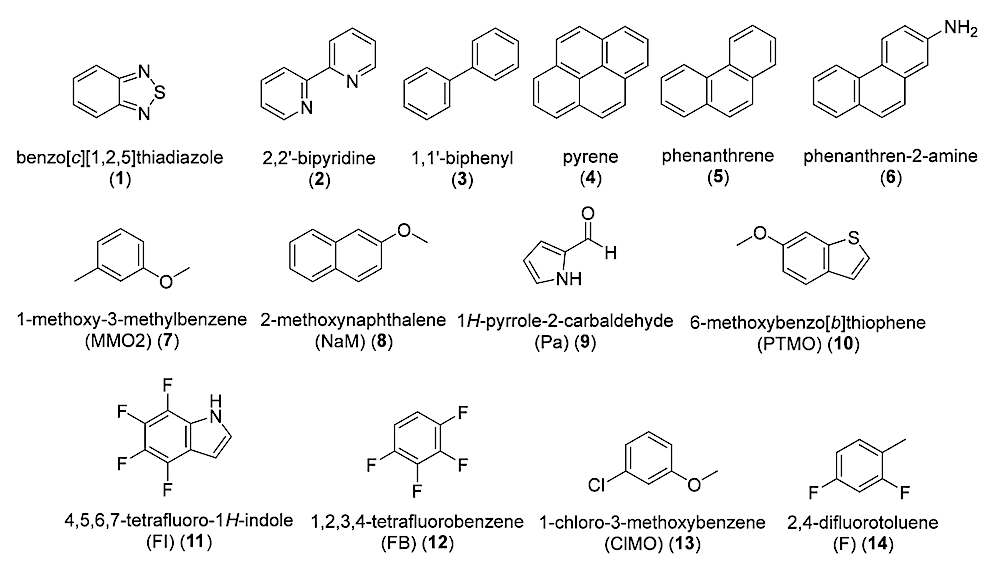
Structures of nucleic acid bases studied in concentrated sulfuric acid in this work- Molecules via PubMed.
We incubate the selected compounds in 98% and 81% w/w sulfuric acid and monitor their stability using 1H and 13C NMR spectroscopy over 3 weeks at room temperature. In 98% w/w sulfuric acid, six bases—benzo[c][1,2,5]thiadiazole (1), 2,2′-bipyridine (2), 1,1′-biphenyl (3), 1-methoxy-3-methylbenzene (MMO2) (7) and 1-chloro-3-methoxybenzene (ClMO) (13), and 2,4-difluorotoluene (14)—remain soluble and stable with no detectable degradation.
A few compounds show non-destructive reactivity, like sulfonation (compound 3) or H/D exchange (compounds 7, 13, 14). The other compounds react rapidly or are insoluble in 98% w/w sulfuric acid. In 81% w/w sulfuric acid, only compounds 1 and 2 remain stable and soluble, while other selected compounds are insoluble or unstable.
Our findings identify a subset of alternative bases stable in concentrated sulfuric acid, advancing efforts towards the design of an example genetic-like polymer in this unusual solvent.
Our work further highlights sulfuric acid’s potential for supporting complex organic chemistry, with implications for astrobiology, planetary science of Venus and synthetic biology.
- Stability and Reactivity of Alternative Nucleobases in Concentrated Sulfuric Acid, Molecules via PubMed (open access)
- Stability and Reactivity of Alternative Nucleobases in Concentrated Sulfuric Acid, Molecules (open access)
Astrobiology,