Ancestors Of Prochlorococcus Microbes Rode Out To Sea On Exoskeleton Particles
Throughout the ocean, billions upon billions of plant-like microbes make up an invisible floating forest. As they drift, the tiny organisms use sunlight to suck up carbon dioxide from the atmosphere.
Collectively, these photosynthesizing plankton, or phytoplankton, absorb almost as much CO2 as the world’s terrestrial forests. A measurable fraction of their carbon-capturing muscle comes from Prochlorococcus — an emerald-tinged free-floater that is the most abundant phytoplankton in the oceans today.
But Prochlorococcus didn’t always inhabit open waters. Ancestors of the microbe likely stuck closer to the coasts, where nutrients were plentiful and organisms survived in communal microbial mats on the seafloor. How then did descendants of these coastal dwellers end up as the photosynthesizing powerhouses of the open oceans today?
MIT scientists believe that rafting was the key. In a new study they propose that ancestors of Prochlorococcus acquired an ability to latch onto chitin — the degraded particles of ancient exoskeletons. The microbes hitched a ride on passing flakes, using the particles as rafts to venture further out to sea. These chitin rafts may have also provided essential nutrients, fueling and sustaining the microbes along their journey.
Thus fortified, generations of microbes may have then had the opportunity to evolve new abilities to adapt to the open ocean. Eventually, they would have evolved to a point where they could jump ship and survive as the free-floating ocean dwellers that live today.
“If Prochlorococcus and other photosynthetic organisms had not colonized the ocean, we would be looking at a very different planet,” says Rogier Braakman, a research scientist in MIT’s Department of Earth, Atmospheric, and Planetary Sciences (EAPS). “It was the fact they were able to attach to these chitin rafts that enabled them to establish a foothold in an entirely new and massive part of the planet’s biosphere, in a way that changed the Earth forever.”
Braakman and his collaborators present their new “chitin raft” hypothesis, along with experiments and genetic analyses supporting the idea, in a study appearing this week in PNAS.
MIT co-authors are Giovanna Capovilla, Greg Fournier, Julia Schwartzman, Xinda Lu, Alexis Yelton, Elaina Thomas, Jack Payette, Kurt Castro, Otto Cordero, and MIT Institute Professor Sallie (Penny) Chisholm, along with colleagues from multiple institutions including the Woods Hole Oceanographic Institution.
A strange gene
Prochlorococcus is one of two main groups belonging to a class known as picocyanobacteria, which are the smallest photosynthesizing organisms on the planet. The other group is Synechococcus, a closely related microbe that can be found abundantly in ocean and freshwater systems. Both organisms make a living through photosynthesis.
But it turns out that some strains of Prochlorococcus can adopt alternative lifestyles, particularly in low-lit regions where photosynthesis is difficult to maintain. These microbes are “mixotrophic,” using a mix of other carbon-capturing strategies to grow.
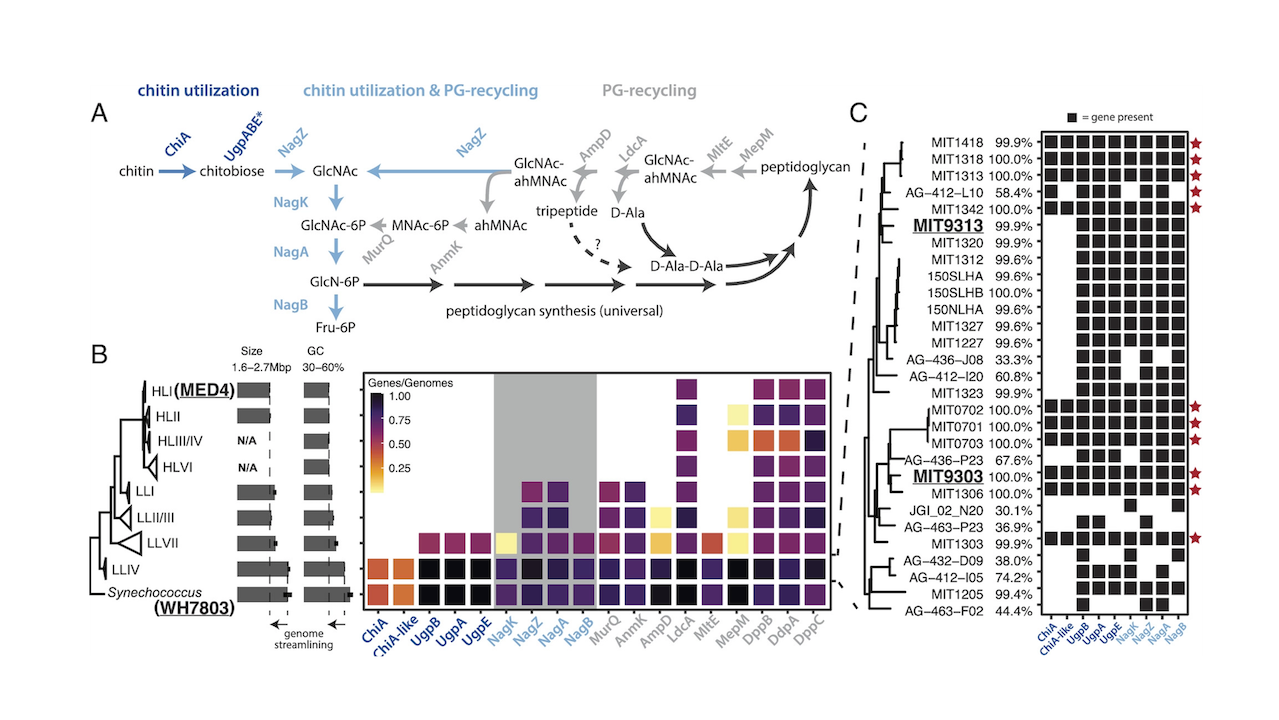
Researchers in Chisholm’s lab were looking for signs of mixotrophy when they stumbled on a common gene in several modern strains of Prochlorococcus. The gene encoded the ability to break down chitin, a carbon-rich material that comes from the sloughed-off shells of arthropods, such as insects and crustaceans.
“That was very strange,” says Capovilla, who decided to dig deeper into the finding when she joined the lab as a postdoc.
For the new study, Capovilla carried out experiments to see whether Prochlorococcus can in fact break down chitin in a useful way. Previous work in the lab showed that the chitin-degrading gene appeared in strains of Prochlorococcus that live in low-light conditions, and in Synechococcus. The gene was missing in Prochlorococcus inhabiting more sunlit regions.
In the lab, Capovilla introduced chitin particles into samples of low-light and high-light strains. She found that microbes containing the gene could degrade chitin, and of these, only low-light-adapted Prochlorococcus seemed to benefit from this breakdown, as they appeared to also grow faster as a result. The microbes could also stick to chitin flakes — a result that particularly interested Braakman, who studies the evolution of metabolic processes and the ways they have shaped the Earth’s ecology.
“People always ask me: How did these microbes colonize the early ocean?” he says. “And as Gio was doing these experiments, there was this ‘aha’ moment.”
Braakman wondered: Could this gene have been present in the ancestors of Prochlorococcus, in a way that allowed coastal microbes to attach to and feed on chitin, and ride the flakes out to sea?
It’s all in the timing
To test this new “chitin raft” hypothesis, the team looked to Fournier, who specializes in tracing genes across species of microbes through history. In 2019, Fournier’s lab established an evolutionary tree for those microbes that exhibit the chitin-degrading gene. From this tree, they noticed a trend: Microbes start using chitin only after arthropods become abundant in a particular ecosystem.
For the chitin raft hypothesis to hold, the gene would have to be present in ancestors of Prochlorococcus soon after arthropods began to colonize marine environments.
The team looked to the fossil record and found that aquatic species of arthropods became abundant in the early Paleozoic, about half a billion years ago. According to Fournier’s evolutionary tree, that also happens to be around the time that the chitin-degrading gene appears in common ancestors of Prochlorococcus and Synecococchus.
“The timing is quite solid,” Fournier says. “Marine systems were becoming flooded with this new type of organic carbon in the form of chitin, just as genes for using this carbon spread across all different types of microbes. And the movement of these chitin particles suddenly opened up the opportunity for microbes to really make it out to the open ocean.”
The appearance of chitin may have been especially beneficial for microbes living in low-light conditions, such as along the coastal seafloor, where ancient picocyanobacteria are thought to have lived. To these microbes, chitin would have been a much-needed source of energy, as well as a way out of their communal, coastal niche.
Braakman says that once out at sea, the rafting microbes were sturdy enough to develop other ocean-dwelling adaptations. Millions of years later, the organisms were then ready to “take the plunge” and evolve into the free-floating, photosynthesizing Prochlorococcus that exist today.
“In the end, this is about ecosystems evolving together,” Braakman says. “With these chitin rafts, both arthropods and cyanobacteria were able to expand into the open ocean. Ultimately, this helped to seed the rise of modern marine ecosystems.”
This research was supported by the Simons Foundation, the EMBO Long-Term Fellowship, and by the Human Frontier Science Program. This paper is a contribution from the Simons Collaboration on Ocean Processes and Ecology (SCOPE).
Paper: “Chitin utilization by marine picocyanobacteria and the evolution of a planktonic lifestyle”, PNAS (open access)
Astrobiology