Prebiotic Vitamin B3 Synthesis in Carbonaceous Planetesimals
Aqueous chemistry within carbonaceous planetesimals is promising for synthesizing prebiotic organic matter essential to all life. Meteorites derived from these planetesimals delivered these life building blocks to the early Earth, potentially facilitating the origins of life.
Here, we studied the formation of vitamin B3 as it is an important precursor of the coenzyme NAD(P)(H), which is essential for the metabolism of all life as we know it.
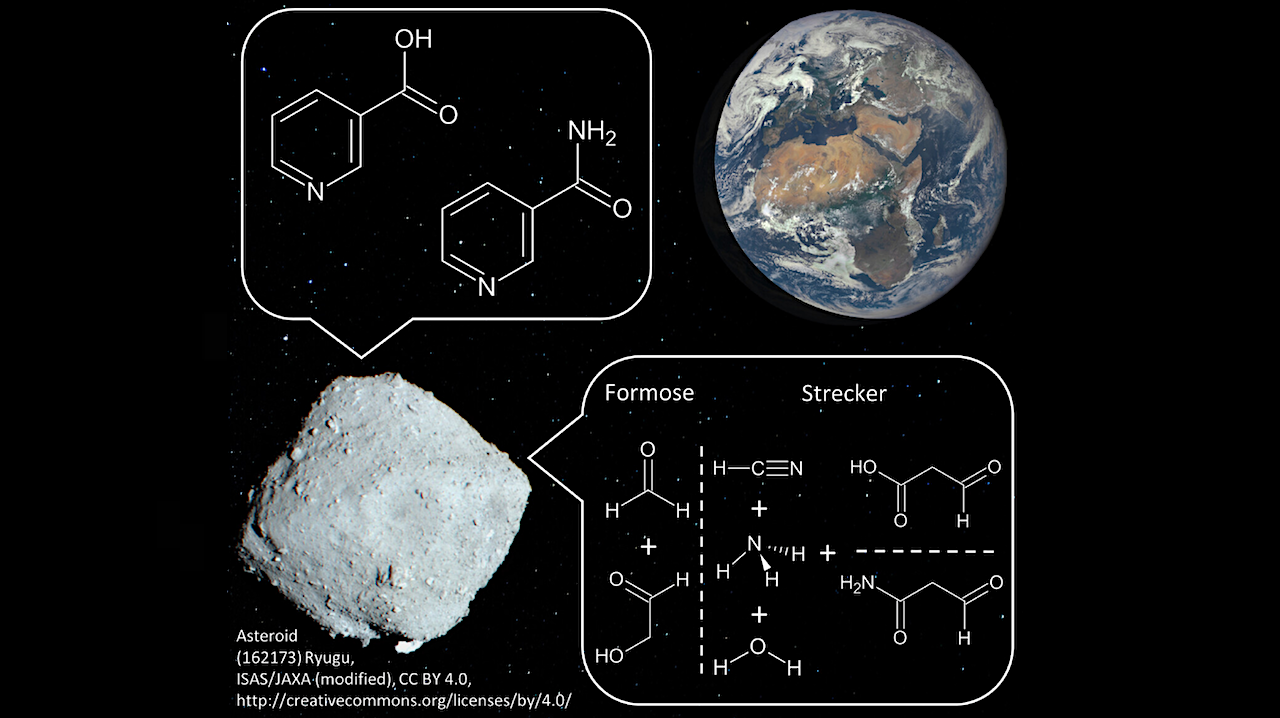
We propose a new reaction mechanism based on known experiments in the literature that explains the synthesis of vitamin B3. It combines the sugar precursors glyceraldehyde or dihydroxyacetone with the amino acids aspartic acid or asparagine in aqueous solution without oxygen or other oxidizing agents.
We performed thermochemical equilibrium calculations to test the thermodynamic favorability. The predicted vitamin B3 abundances resulting from this new pathway were compared with measured values in asteroids and meteorites.
We conclude that competition for reactants and decomposition by hydrolysis are necessary to explain the prebiotic content of meteorites. In sum, our model fits well into the complex network of chemical pathways active in this environment.

Newly proposed reaction mechanism inspired by experimental studies by Cleaves & Miller (2001).79 The phosphate and associated reaction steps are shown in red to indicate that phosphorylation increased the yield in the experiments; however, the synthesis of nicotinic acid was successful even without it, indicating it might be optional. Phosphates might not be readily available in meteorite parent bodies, but the reaction mechanism might still operate. The amide group serves as an alternative to the carboxyl group and is therefore also highlighted in red. Depending on the side chain of the starting aldehyde, either nicotinic acid or nicotinamide is formed in the Strecker synthesis.– ChemPlusChem
Prebiotic Vitamin B3 Synthesis in Carbonaceous Planetesimals, ChemPlusChem (open access)
Astrobiology, Astrochemistry