The Origins Of ATP Dependence In Biological Nitrogen Fixation
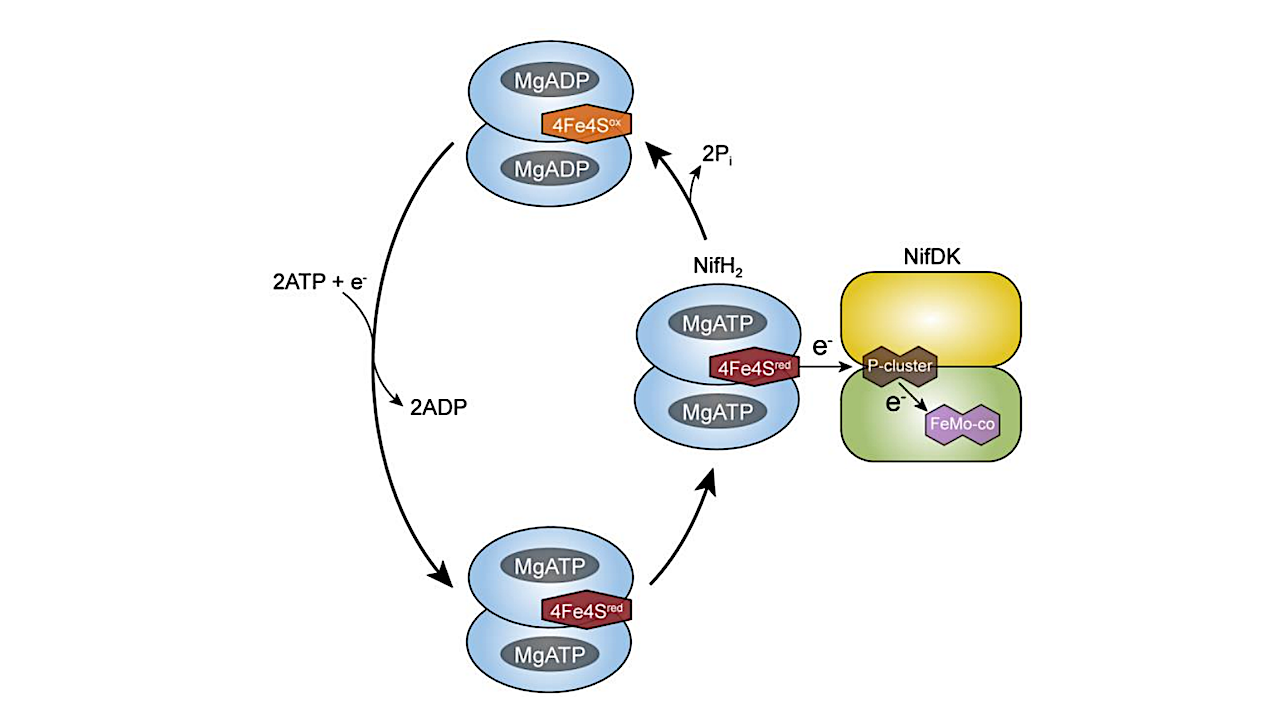
Life depends on a conserved set of chemical energy currencies that are relics of early biochemistry. One of these is ATP, a molecule that, when paired with a divalent metal ion such as Mg2+, can be hydrolyzed to support numerous cellular and molecular processes.
Despite its centrality to extant biochemistry, it is unclear whether ATP supported the function of ancient enzymes. We investigate the evolutionary necessity of ATP by experimentally reconstructing an ancestral variant of the key N2 reducing enzyme nitrogenase. We show that the ancestor has a strict requirement for ATP and its hydrolysis is coupled to electron transfer for N2 reduction.
Our results provide direct laboratory evidence of ATP usage by an ancient enzyme, and underscore how biomolecular constraints can entirely decouple cofactor selection from environmental availability.
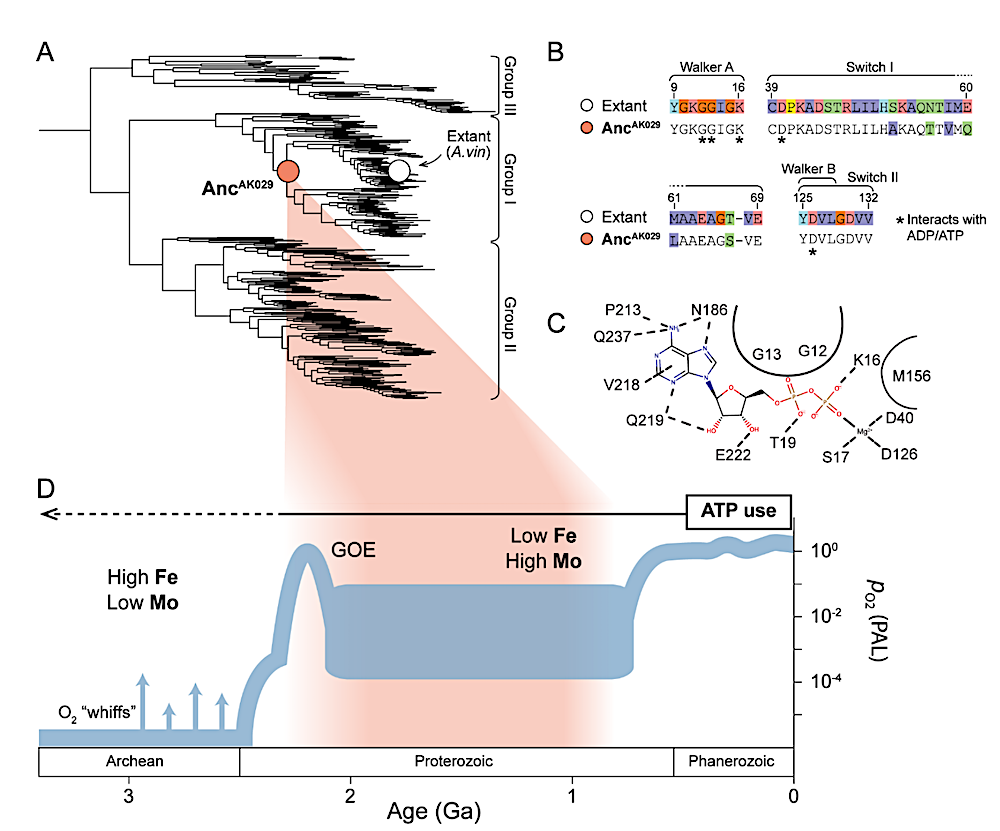
Evolution of the ATP/ADP-binding site in nitrogenase NifH. (A) Nitrogenase NifHDK protein phylogeny. Phylogenetic positions of ancestral (AncAK029 142 ) and extant A. vinelandii (“Extant 143 A. vin”) nitrogenase variants discussed in the main text are highlighted by colored circles. (B) Protein sequence alignment of NifH ATPase signature motifs (labeled) 144 33 Site index from A. 145 vinelandii NifH. (C) Residue-level intermolecular interactions in the ADP binding site of A. vinelandii NifH (PDB 1FP6). All displayed residues are conserved between AncAK029 146 and WT. 147 Curved lines delineate residues with nonspecific interactions that shape the binding site. (D) 148 Estimated age range of nitrogenase ancestors mapped to Earth environmental history. 149 Atmospheric oxygenation plot and relative marine metal abundances are from Lyons et al. (2014). — biorxiv.org
The Origins of ATP Dependence in Biological Nitrogen Fixation
Derek Harris, Holly Rucker, Amanda Garcia, Zhi-Yong Yang, Scott Chang, Hannah Feinsilber, Betul Kacar, Lance Seefeldt
doi: https://doi.org/10.1101/2024.02.22.581614
Astrobiology