Simulation Of CH3OH Ice UV Photolysis Under Laboratory Conditions
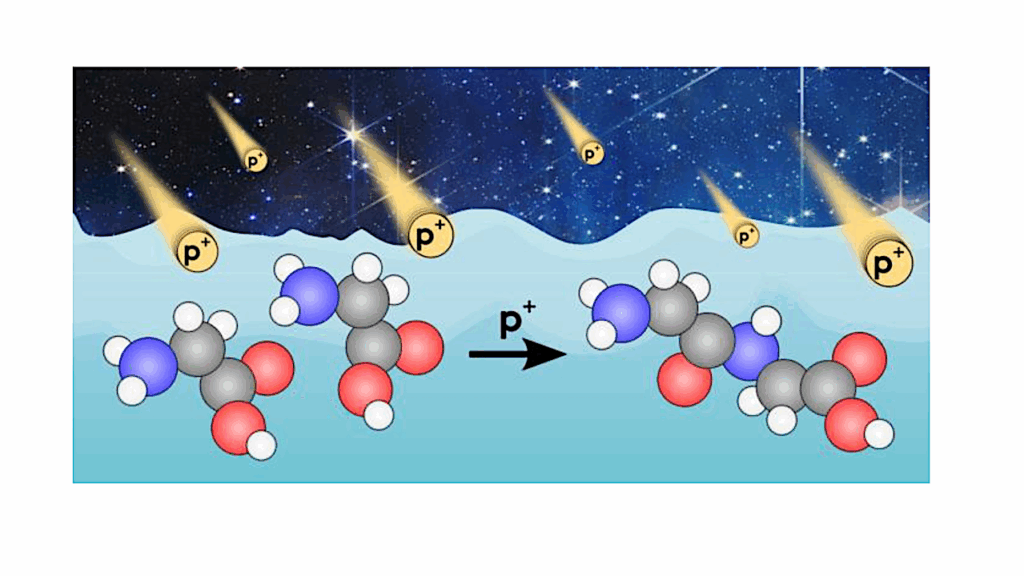
Methanol is the most complex molecule securely identified in interstellar ices and is a key chemical species for understanding chemical complexity in astrophysical environments.
Important aspects of the methanol ice photochemistry are still unclear such as the branching ratios and photo-dissociation cross-sections at different temperatures and irradiation fluxes. This work aims at a quantitative agreement between laboratory experiments and astrochemical modelling of the CH3OH ice UV photolysis. This work also allows us to better understand which processes govern the methanol ice photochemistry present in laboratory experiments.
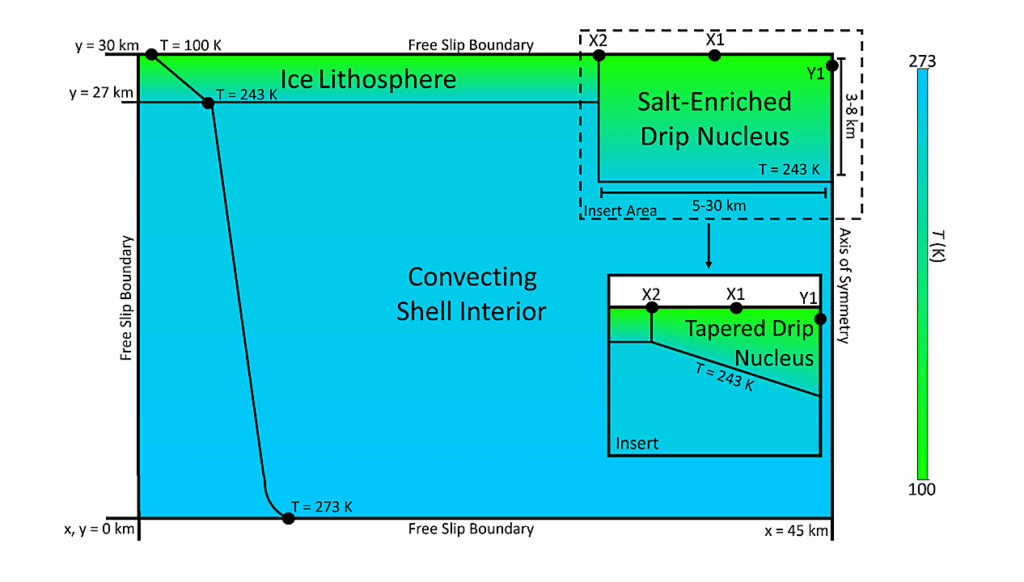
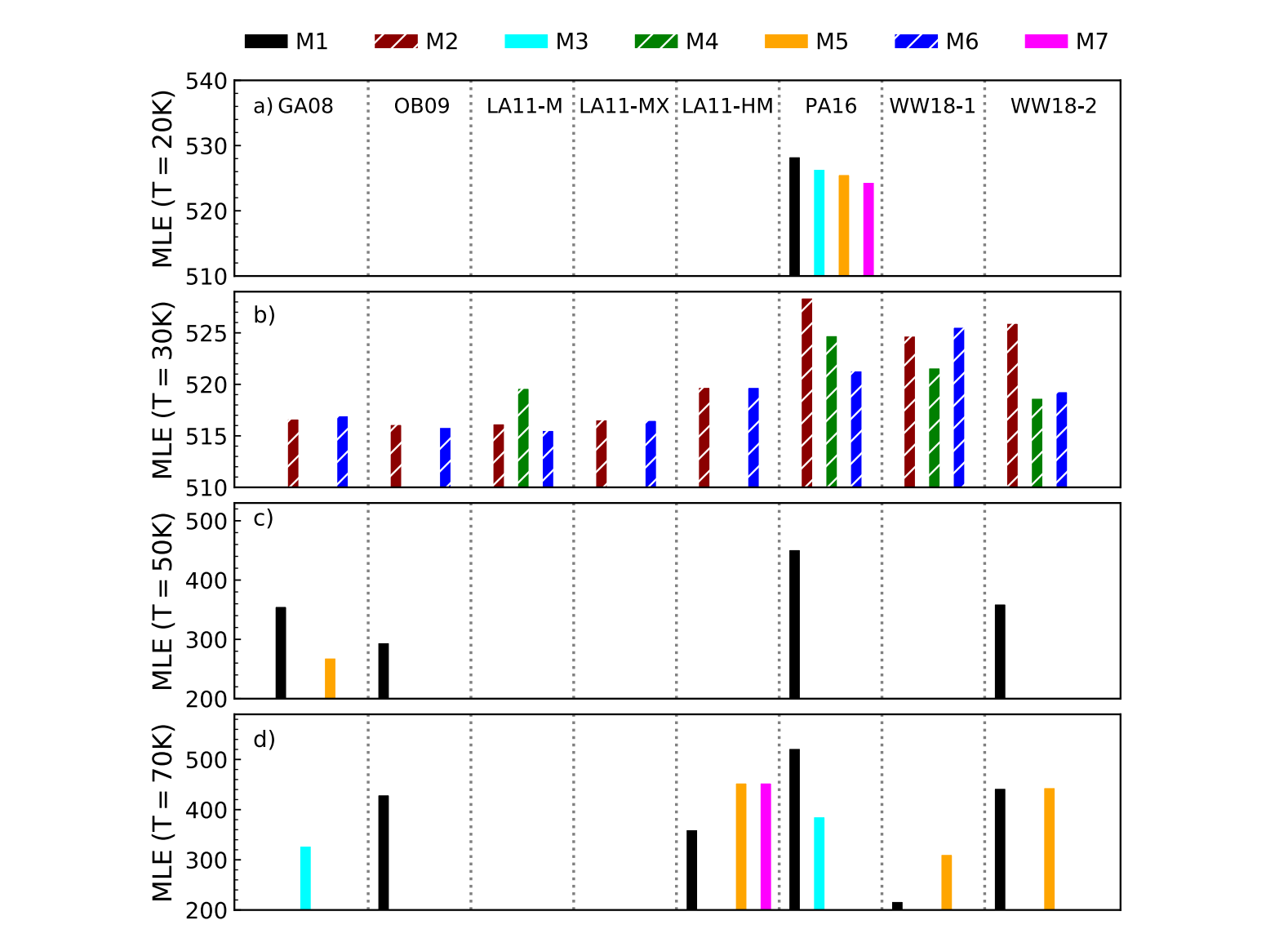
We use ProDiMo to simulate the conditions of laboratory measurements. The simulations start with simple chemistry consisting only of methanol ice and helium to mimic the residual gas in the experimental chamber. A surface chemical network enlarged by photo-dissociation reactions is used to study the chemical reactions within the ice. Additionally, different surface chemistry parameters (surface competition, tunnelling, thermal diffusion and reactive desorption) are adopted to check those that reproduce the experimental results. The chemical models with ProDiMo can reproduce the methanol ice destruction via UV photodissociation at temperatures of 20, 30, 50 and 70 K as observed in the experiments.
We note that the results are sensitive to different branching ratios after photolysis and to the mechanisms of reactive desorption. In the simulations of a molecular cloud at 20 K, we observed an increase in the methanol gas abundance of one order of magnitude, with a similar decrease in the solid-phase abundance. Comprehensive astrochemical models provide new insights into laboratory experiments as the quantitative understanding of the processes that govern the reactions within the ice. Ultimately, those insights can help to better interpret astronomical observations.
W. R. M. Rocha, P. Woitke, S. Pilling, W. -F. Thi, J. K. Jørgensen, L. E. Kristensen, G. Perotti, I. Kamp
Comments: Accepted for publication in A&A on 08-Feb-2023
Subjects: Astrophysics of Galaxies (astro-ph.GA); Solar and Stellar Astrophysics (astro-ph.SR); Chemical Physics (physics.chem-ph)
Cite as: arXiv:2302.04239 [astro-ph.GA] (or arXiv:2302.04239v1 [astro-ph.GA] for this version)
Submission history
From: Will Robson Monteiro Rocha
[v1] Wed, 8 Feb 2023 18:09:26 UTC (32,914 KB)
https://arxiv.org/abs/2302.04239
Astrobiology, Astrochemistry