Stolen Chloroplasts Maintained By Host-made Proteins Offer Clues To Plant Cell Origins
Every plant cell is the product of a biological merger billions of years ago. Chloroplasts are key structures in plants and algae that capture sunlight, but originally they were free-living bacteria that took up residence inside another cell. Over time, these partners became more closely integrated by sharing genes, proteins, and roles.
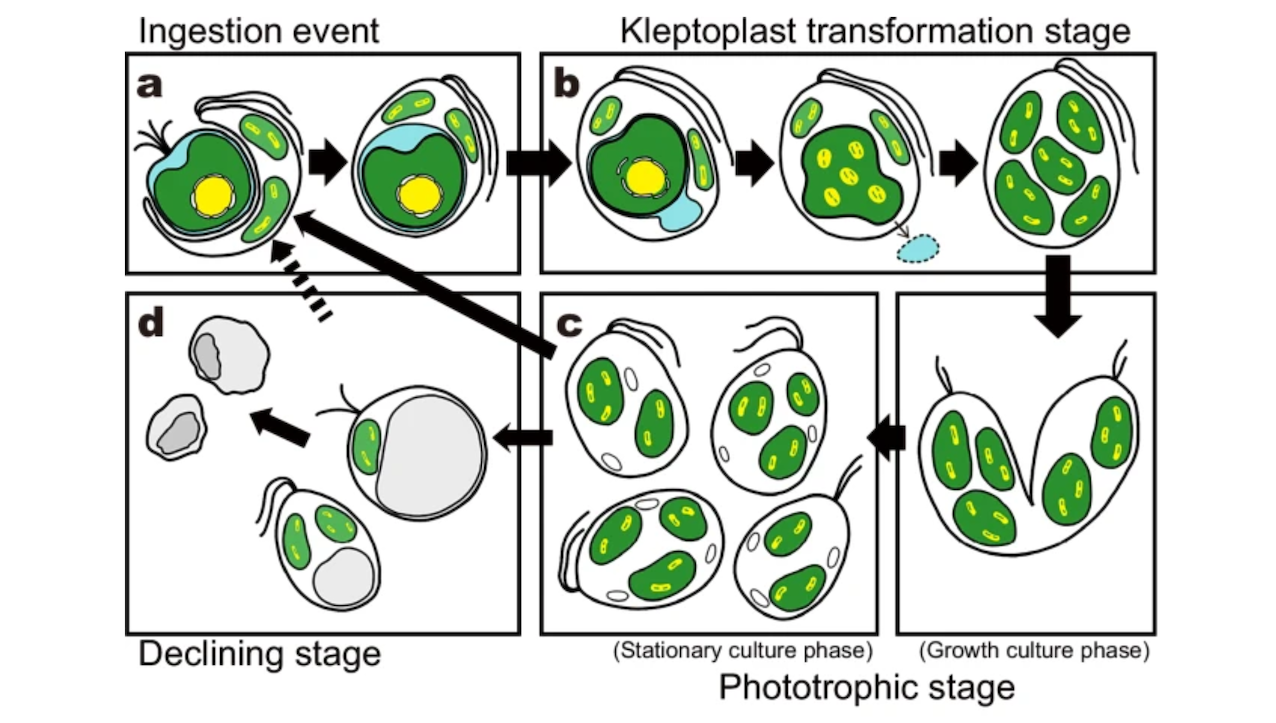
To understand how this process happened, scientists look for organisms that display similar processes. A tiny predator named Rapaza viridis may offer a glimpse of some of the early steps involved in that ancient transformation.
R. viridis is a single-celled organism that performs photosynthesis using chloroplasts stolen from the green alga it consumes. This process is called kleptoplasty—from the Greek word for thief.
Even after the algal nucleus and much of the cytoplasm are lost, the prey-derived chloroplasts remain inside R. viridis. Temporarily, structures from two different organisms coexist within a single cell, which can be described as structural-level chimerism.
Using genetic engineering and biochemical approaches, research led by Masami Nakazawa, a lecturer at the Graduate School of Agriculture, Osaka Metropolitan University, and Professor Yuichiro Kashiyama at the Faculty of Environmental Studies, Fukui University of Technology, has found that the kleptoplasts in R. viridis also exhibit chimerism at the molecular level. This suggests a more advanced form of kleptoplasty than structural-level chimerism alone.
The group identified host-made proteins that are transported into the stolen chloroplast, where they help keep key chloroplast machinery working. When the researchers disrupted the genes encoding these proteins, the stolen chloroplasts functioned less effectively.
Their findings suggest that by producing proteins that function within the stolen chloroplast, R. viridis goes beyond simple prey retention and offers a valuable model for studying how deeper host–organelle integration can arise.
“This makes R. viridis the first organism in which proteins encoded in the host’s nucleus have been biochemically shown to function inside a stolen organelle from another species,” Professor Kashiyama said. “These findings show that even temporary chloroplast retention can involve a deeper level of host–organelle integration than previously recognized.”
Dr. Nakazawa believes that experiments like these can help researchers understand what happened when the first plant cells emerged. “By revealing mechanisms at work when eukaryotic cells use foreign organelles, going beyond what we typically see in model organisms, this study provides clues to the evolutionary processes that gave rise to plant cells,” she said.
Transient molecular chimerism for exploiting xenogeneic organelles, Nature Communications (open access)
Astrobiology