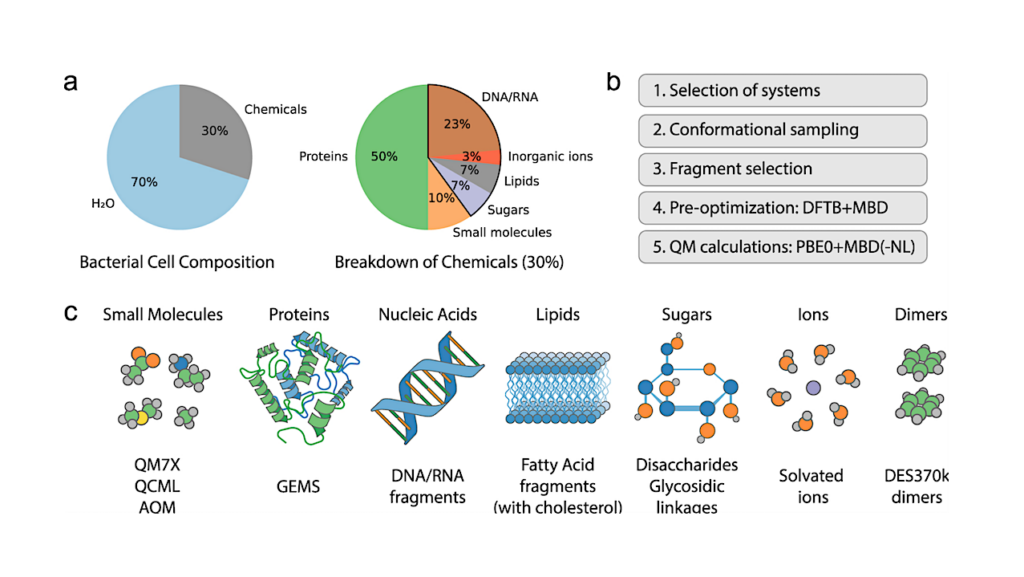
ExocubeBio: An In-situ Fluidic Platform For Microbial Exposure On The International Space Station

Space-based platforms currently represent the most accurate means to experimentally assess the influence of the space environment on biological systems. However, performing such experiments remains technically challenging and requires highly specialized instrumentation.
This study describes the current development and hardware qualification of ExocubeBio, a miniaturized experimental platform for in-situ biological space exposure. This experiment is scheduled for installation on the exterior of the International Space Station in 2027, as part of Exobio, the European Space Agency’s new generation exobiology exposure facility.
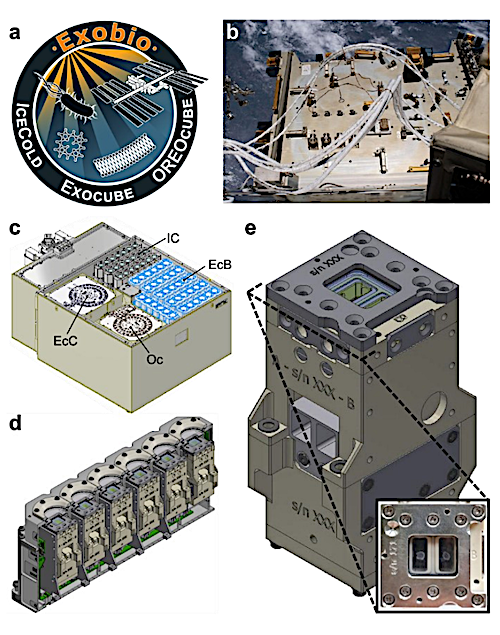
ExocubeBio design overview. (a) The Exobio project logo. (b) Photograph of the Bartolomeo science platform, outside of the ISS Columbus module (image credit: ESA/NASA, 2021 [28]). CAD illustrations depicting (c) the Exobio science module with hardware of each experiment labeled as follows: EcB = ExocubeBio (highlighted in blue), EcC = ExocubeChem, IC = IceCold, Oc = OREOcube, (d) a single ExocubeBio cartridge, shown in cross section to reveal the six experimental units contained inside, and e. a single ExocubeBio experimental unit. The inset is a top-down photo of an experimental unit, showing two independently housed, dry microbial samples deposited beneath the MgF2 window. — biorxiv.org
ExocubeBio will expose live microbial samples to the low Earth orbit environment, and combine autonomous in-situ optical density and fluorescence measurements, with the capacity to return preserved samples to Earth. Achieving these experimental goals requires a specialized, robust and reliable hardware system.
The ExocubeBio hardware testing described here includes assessment of material biocompatibility and durability, functional validation of the miniaturized fluidic system, and optimization of the integrated optical subsystem for optical density and fluorescence measurements.
These results demonstrate that the ExocubeBio experimental hardware components can each execute their core functional and operational requirements; subsystems allow for sample exposure, in-situ measurements of microbial cultures, and the chemical preservation of samples for post-flight analysis.
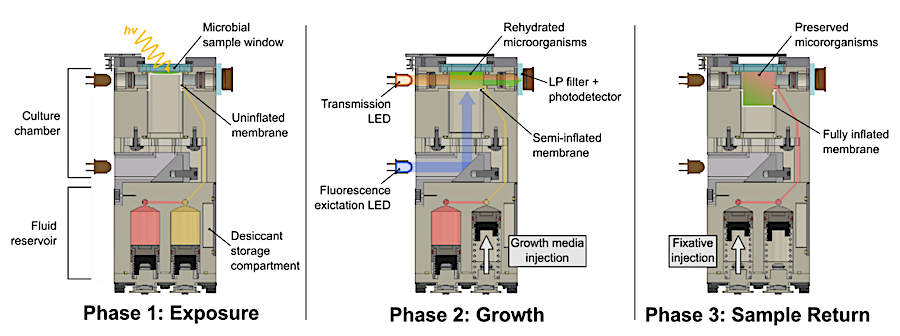
CAD diagrams illustrating the intended hardware function at each ExocubeBio experimental phase. As these diagrams are conceptual examples, some elements have been visually simplified or omitted for legibility. Illustrative elements or those not visible in this diagram include the liquid transport channels that run perpendicular to the plane of view shown, thermal plates housed on either side of each experimental unit, and air lines that connect the desiccant storage compartment to the gaseous headspace beneath the silicone membrane. — biorxiv.org
As ExocubeBio transitions from hardware development to mission readiness, the results presented here validate the overall design and engineering approaches utilized. By combining the strengths of in-situ monitoring and sample return, ExocubeBio represents a significant advancement in space-based experimentation, and will provide new insights into microbial responses to the space environment.

ExocubeBio fluidic system functionality. (a) Mean relative humidity measured over time, within Exocube culture chambers (n = 2, biological replicates). The nominal hardware configuration (orange) is compared against experimental units prepared with a perforation in the silicone membrane between the culture chamber and the liquid transport channels (blue), and a unit prepared without liquids and left open to the dry, nitrogen atmosphere (yellow). Shaded regions represent standard deviations plus humidity sensor accuracy specifications. (b) Photos of liquid-filled culture chambers of an ExocubeBio experimental unit, shown from top-down (left) and through the optical pathway (right). The upper pair of photos demonstrate a nominal activation of the fluidics system, completely filling the culture chamber. In contrast, the lower pair of photos are a representative example of a partial experimental failure, due to large bubbles within the culture chamber, indicative of incorrect filling. (c) TEM imagining of C. reinhardtii cells preserved with different fixative solutions, including four concentrations of aldehydes (4.5% control – 0%), undiluted and a 50% dilution of the aldehyde-free fixative NOTOXhisto (NTH), and standard aldehyde-based fixative solutions exposed to simulated ISS-level, annual doses of either gamma or proton irradiation. Labeled cellular features are as follows: Nu = nucleus (within the nucleolus), PM = plasma membrane, Py = pyrenoid, SG = starch granule, TM = thylakoid membrane. Scale bars equal 1 µm. — biorxiv.org
ExocubeBio: an in-situ fluidic platform for microbial exposure on the International Space Station, biorxiv.org
Astrobiology, space biology, microgravity,