Emergence Of A Potentially Ancestral ATP-synthesizing Activity Under Prebiotic Amino Acid Constraints
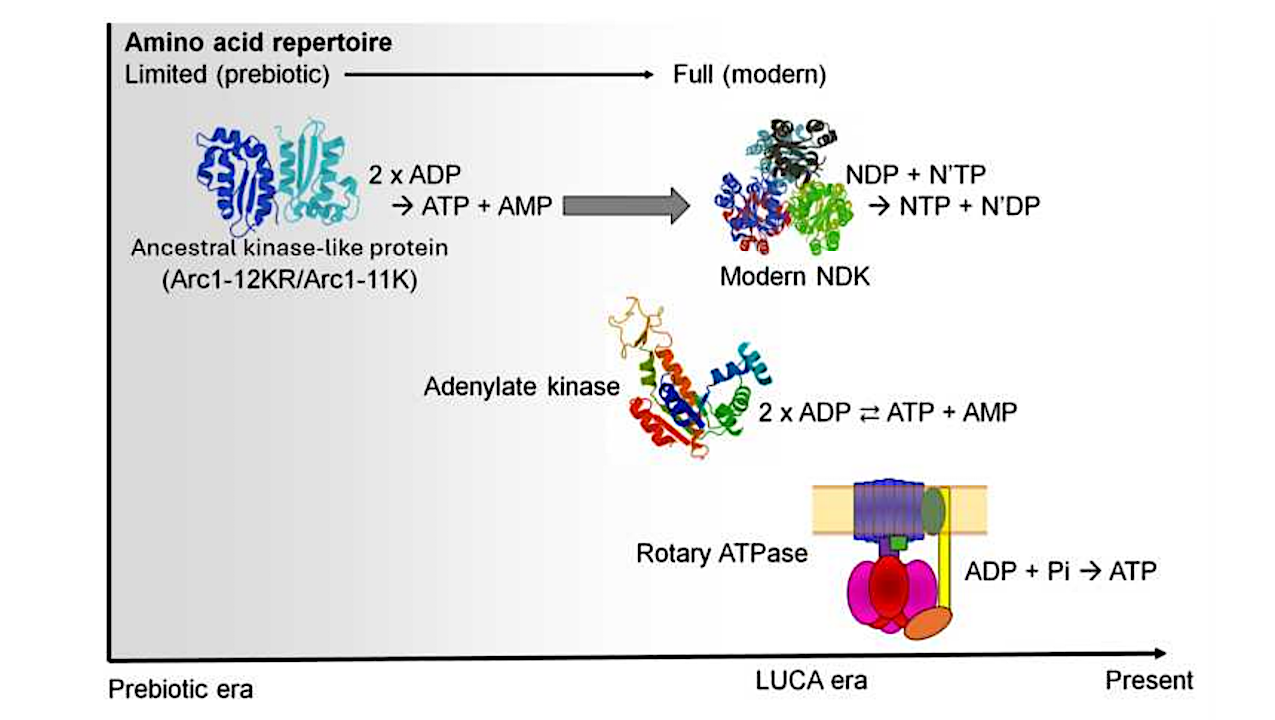
The earliest enzymes are thought to have been composed of the limited amino acid repertoire available on prebiotic Earth. However, how such compositional simplicity shaped catalytic capability remains unclear.
Here, Arc1-12KR, a variant of an ancestral nucleoside diphosphate kinase (NDK), was reconstructed using a restricted amino acid alphabet of ten prebiotic amino acids plus lysine and arginine. Arc1-12KR catalyzes the disproportionation of two ADP molecules into ATP and AMP, a reaction not observed in extant NDKs but chemically identical to that of adenylate kinase, suggesting a convergence on a primordial ATP-generating chemistry.
Although modest in catalytic rate, X-ray crystallography, molecular dynamics, and mutational analyses indicate that aspartate and arginine cooperatively coordinate Mg2+ and organize the active site in a manner distinct from canonical NDKs, enabling ATP formation through a noncanonical phosphoryl-transfer mechanism.
Moreover, a variant in which all arginine residues were replaced with lysine retained substantial ATP-synthesizing activity, indicating that lysine can substitute for arginine under such constraints. Together, these findings show that restricted amino acid availability can remodel active sites and promote noncanonical ATP-generating phosphoryl-transfer chemistry, offering a plausible path for the early evolution of enzymatic ATP production.
Emergence of a potentially ancestral ATP-synthesizing activity under prebiotic amino acid constraints, biorxiv.org
Astrobiology,