Binding Energies of Ethanol and Ethylamine on Interstellar Water Ices: Synergy Between Theory and Experiments
Experimental and computational chemistry are two disciplines to conduct research in Astrochemistry, providing essential reference data for both astronomical observations and modeling. These approaches not only mutually support each other, but also serve as complementary tools to overcome their respective limitations.
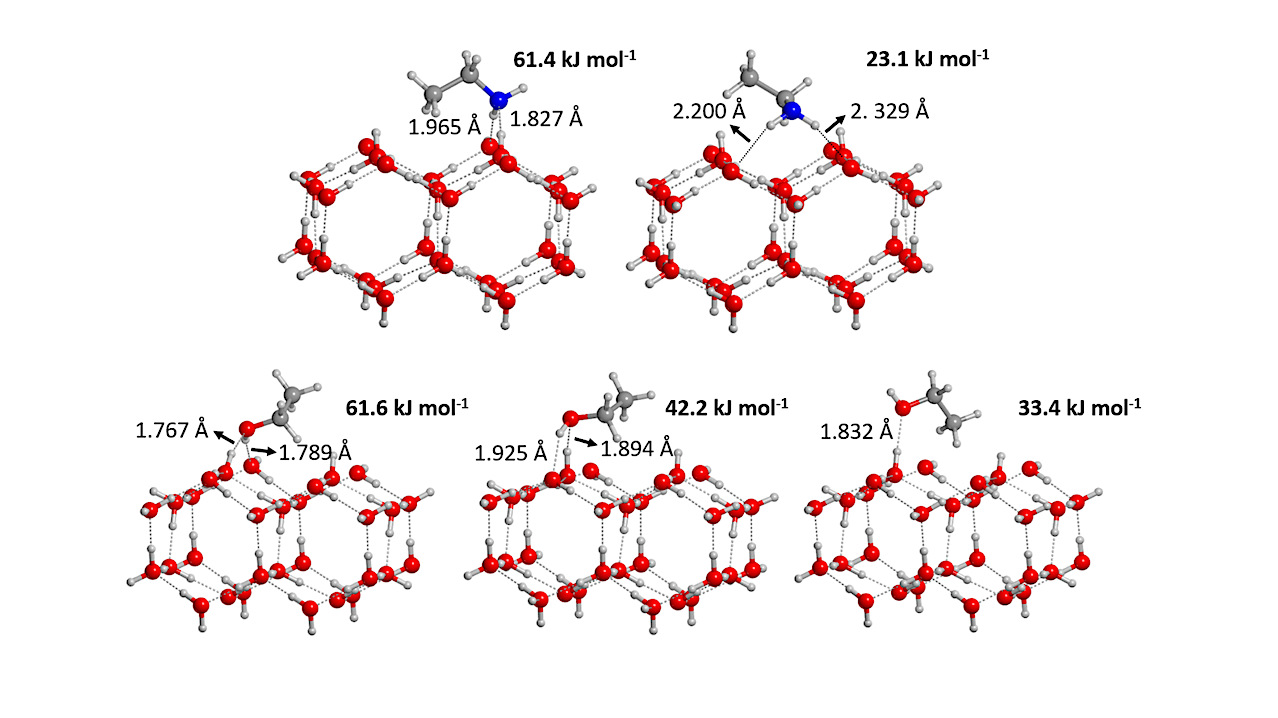
We characterized the binding energies (BEs) of ethanol (CH3CH2OH) and ethylamine (CH3CH2NH2), two interstellar complex organic molecules (iCOMs), onto crystalline and amorphous water ices through density functional theory (DFT) calculations and temperature programmed desorption (TPD) experiments. Experimentally, CH3CH2OH and CH3CH2NH2 behave similarly, in which desorption temperatures are higher on the water ices than on a bare gold surface.
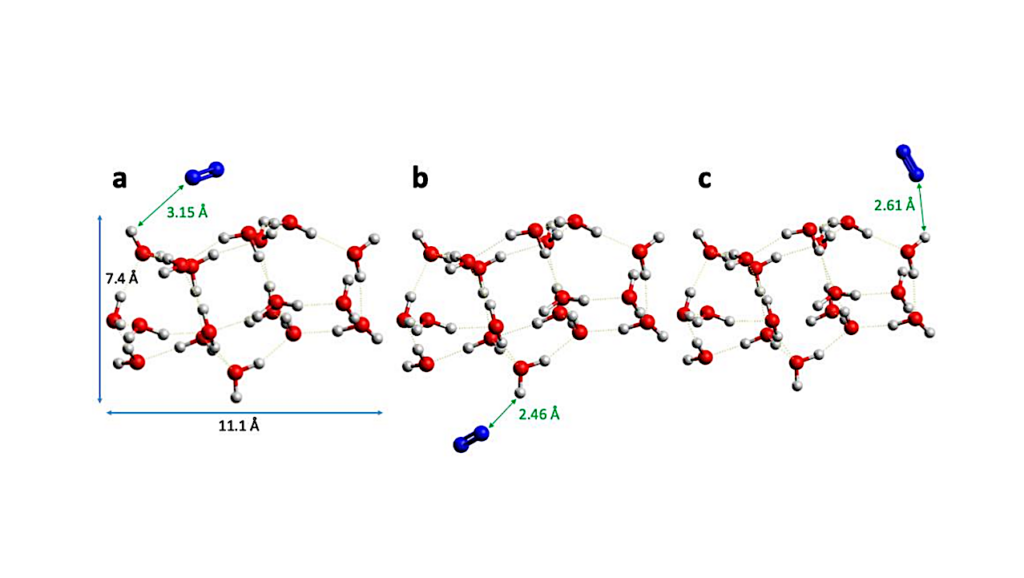
Computed cohesive energies of pure ethanol and ethylamine bulk structures allow describing the BEs of the pure species deposited on the gold surface, as extracted from the TPD curve analyses. The BEs of submonolayer coverages of CH3CH2OH and CH3CH2NH22 on the water ices cannot be directly extracted from TPD due to their co-desorption with water, but they are computed through DFT calculations, and found to be greater than the cohesive energy of water.
The behaviour of CH3CH2OH and CH3CH2NH2 is different when depositing adsorbate multilayers on the amorphous ice, in that, according to their computed cohesive energies, ethylamine layers present weaker interactions compared to ethanol and water.
Finally, from the computed BEs of ethanol, ethylamine and water, we can infer that the snow-lines of these three species in protoplanetary disks will be situated at different distances from the central star. It appears that a fraction of ethanol and ethylamine is already frozen on the grains in the water snow-lines, causing their incorporation in water-rich planetesimals.
Jessica Perrero, Julie Vitorino, Emanuele Congiu, Piero Ugliengo, Albert Rimola, François Dulieu
Comments: 18 pages, 7 figures, accepted for publication in PCCP
Subjects: Chemical Physics (physics.chem-ph); Astrophysics of Galaxies (astro-ph.GA)
Cite as: arXiv:2406.16952 [physics.chem-ph] (or arXiv:2406.16952v1 [physics.chem-ph] for this version)
Related DOI:
https://doi.org/10.1039/D4CP01934B
Focus to learn more
Submission history
From: Jessica Perrero
[v1] Thu, 20 Jun 2024 14:17:21 UTC (4,188 KB)
https://arxiv.org/abs/2406.16952
Astrobiology, Astrochemistry,