Reaction Kinetics Of CN + Toluene And Its Implication On The Productions Of Aromatic Nitriles In The Taurus Molecular Cloud And Titan’s Atmosphere
Reactions between cyano radical and aromatic hydrocarbons are believed to be important pathways for the formation of aromatic nitriles in the interstellar medium (ISM) including those identified in the Taurus molecular cloud (TMC-1).
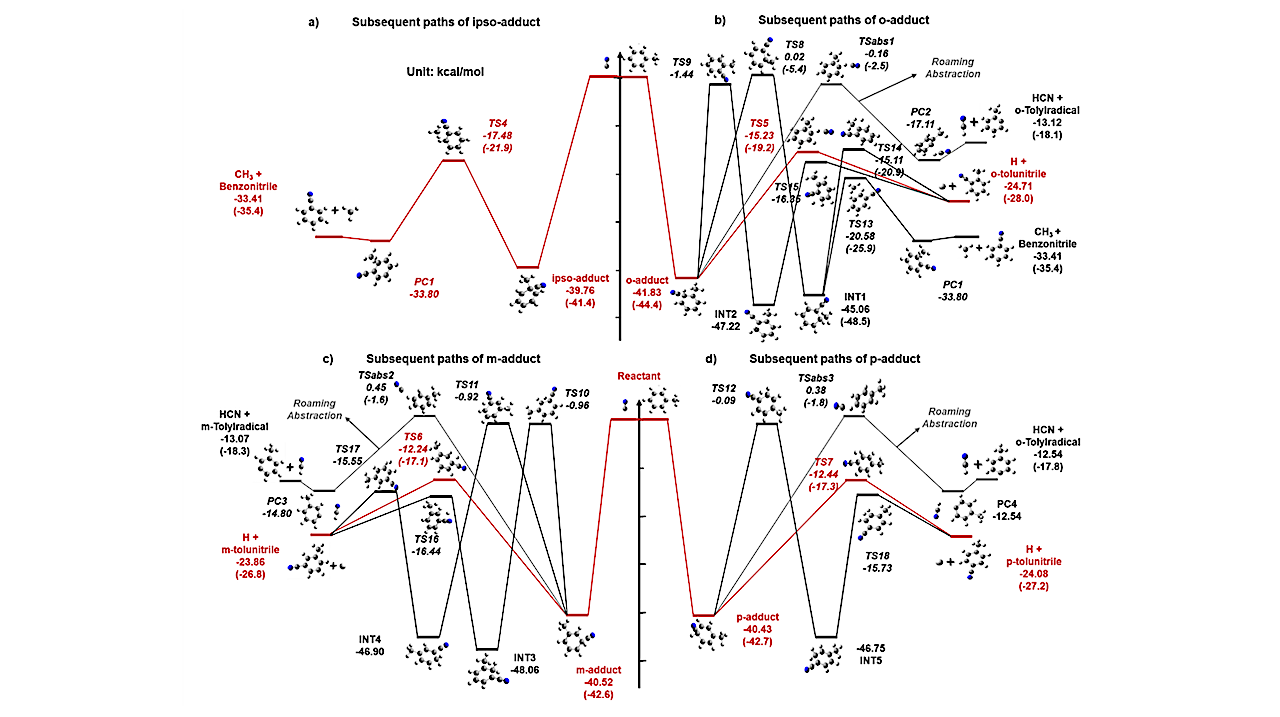
Aromatic nitriles might participate in the formation of polycyclic aromatic nitrogen containing hydrocarbons (PANHs) in Titan’s atmosphere. Here, ab initio kinetics simulations reveal a high efficiency of ∼10−10 cm3 s−1 and the competition of the different products of 30-1800 K and 10−7-100 atm of the CN + toluene reaction.
In the star-forming region of TMC-1 environment, the product yields of benzonitrile and tolunitriles for CN reacting with toluene may be approximately 17% and 83%, respectively. The detection of main products, tolunitriles, can serve as proxies for the undetected toluene in the ISM due to their much larger dipole moments. The competition between bimolecular and unimolecular products is extremely intense under the warmer and denser PANH forming region of Titan’s stratosphere.
The computational results show that the fractions of tolunitriles, adducts, and benzonitrile are 19%-68%, 15%-64% and 17%, respectively, at 150-200 K and 0.0001-0.001 atm (Titan’s stratosphere). Then, benzonitrile and tolunitriles may contribute to the formation of PANHs by consecutive C2H additions. Kinetic information of aromatic nitriles for the CN + toluene reaction calculated here helps to explain the formation mechanism of polycyclic aromatic hydrocarbons (PAHs) or PANHs under different interstellar environments and constrains corresponding astrochemical models.
Mengqi Wu, Xiaoqing Wu, Qifeng Hou, Jiangbin Huang, Dongfeng Zhao, Feng Zhang
Subjects: Astrophysics of Galaxies (astro-ph.GA); Solar and Stellar Astrophysics (astro-ph.SR)
Cite as: arXiv:2304.03582 [astro-ph.GA] (or arXiv:2304.03582v1 [astro-ph.GA] for this version)
Submission history
From: Mengqi Wu
[v1] Fri, 7 Apr 2023 10:48:34 UTC (1,925 KB)
https://arxiv.org/abs/2304.03582
Astrobiology, Astrochemistry