A Newly Discovered Form Of Salty Ice Could Exist On The Surface Of Icy Moons

The red streaks crisscrossing the surface of Europa, one of Jupiter’s moons, are striking. Scientists suspect it is a frozen mixture of water and salts, but its chemical signature is mysterious because it matches no known substance on Earth.
A team led by the University of Washington may have solved the puzzle with the discovery of a new type of solid crystal that forms when water and table salt combine in cold and high-pressure conditions. Researchers believe the new substance created in a lab on Earth could form at the surface and bottom of these worlds’ deep oceans.
The study, published Feb. 20 in the Proceedings of the National Academy of Sciences, announces a new combination for two of Earth’s most common substances: water and sodium chloride, or table salt.
“It’s rare nowadays to have fundamental discoveries in science,” said lead author Baptiste Journaux, a UW acting assistant professor of Earth and space sciences. “Salt and water are very well known at Earth conditions. But beyond that, we’re totally in the dark. And now we have these planetary objects that probably have compounds that are very familiar to us, but in at very exotic conditions. We have to redo all the fundamental mineralogical science that people did in the 1800s, but at high pressure and low temperature. It is an exciting time.”

This satellite image shows white streaks across the surface of Ganymede, the largest of Jupiter’s moons. The discovery of new types of salty ice could explain the material in these streaks and provide clues on the composition of Ganymede’s ice-covered ocean. CREDIT NASA/JPL/JUNO
At cold temperatures water and salts combine to form a rigid salted icy lattice, known as a hydrate, held in place by hydrogen bonds. The only previously known hydrate for sodium chloride was a simple structure with one salt molecule for every two water molecules.
But the two new hydrates, found at moderate pressures and low temperatures, are strikingly different. One has two sodium chlorides for every 17 water molecules; the other has one sodium chloride for every 13 water molecules. This would explain why the signatures from the surface of Jupiter’s moons are more “watery” than expected.
“It has the structure that planetary scientists have been waiting for,” Journaux said.
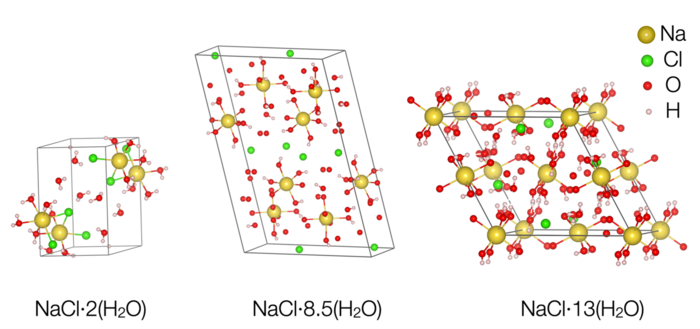
This image shows the newly discovered hydrate that has two sodium chloride molecules for every 17 water molecules. This crystal formed at high pressure but remains stable at cold, low-pressure conditions. CREDIT Journaux et al./PNAS
The discovery of new types of salty ice has importance not just for planetary science, but for physical chemistry and even energy research, which uses hydrates for energy storage, Journaux said.
The experiment involved compressing a tiny bit of salty water between two diamonds about the size of a grain of sand, squeezing the liquid up to 25,000 times the standard atmospheric pressure. The transparent diamonds allowed the team to watch the process through a microscope.
“We were trying to measure how adding salt would change the amount of ice we could get, since salt acts as an antifreeze,” Baptiste said. “Surprisingly, when we put the pressure on, what we saw is that these crystals that we were not expecting started growing. It was a very serendipitous discovery.”
Such cold, high-pressure conditions created in the lab would be common on Jupiter’s moons, where scientists think 5 to 10 kilometers of ice would cover oceans up to several hundred kilometers thick, with even denser forms of ice possible at the bottom.
“Pressure just gets the molecules closer together, so their interaction changes — that is the main engine for diversity in the crystal structures we found,” Journaux said.
Once the newly discovered hydrates had formed, one of the two structures remained stable even after the pressure was released.

Researchers discovered two new crystals made from water and table salt at low temperatures, below about minus 50 C. The known structure (left) has one salt molecule (yellow and green balls) to two water molecules (red and pink balls). X-ray imaging let researchers determine the position of individual atoms in the new structures. The center structure has two sodium chloride molecules for every 17 water molecules and stays stable even if pressure drops to near vacuum, as would exist on a lunar surface. The structure on the right has one sodium chloride molecule for every 13 water molecules, and is stable only at high pressure. CREDIT Baptiste Journaux/University of Washington
“We determined that it remains stable at standard pressure up to about minus 50 C. So if you have a very briny lake, for example in Antarctica, that could be exposed to these temperatures, this newly discovered hydrate could be present there,” Journaux said.
The team hopes to either make or collect a larger sample to allow more thorough analysis and verify whether the signatures from icy moons match the signatures from the newly discovered hydrates.
Two upcoming missions will explore Jupiter’s icy moons: The European Space Agency’s Jupiter Icy Moons Explorer mission, launching in April, and NASA’s Europa Clipper mission, launching for October 2024. NASA’s Dragonfly mission launches to Saturn’s moon Titan in 2026. Knowing what chemicals these missions will encounter will help to better target their search for signatures of life.
“These are the only planetary bodies, other than Earth, where liquid water is stable at geological timescales, which is crucial for the emergence and development of life,” Journaux said. “They are, in my opinion, the best place in our solar system to discover extraterrestrial life, so we need to study their exotic oceans and interiors to better understand how they formed, evolved and can retain liquid water in cold regions of the solar system, so far away from the sun.”
This research was funded by NASA. Co-authors are professor J. Michael Brown and graduate student Jason Ott at the UW. Additional co-authors were at the German Electron Synchrotron in Hamburg; the European Synchrotron Facility in France; the Institute of Geochemistry and Petrology in Switzerland, the Bavarian Geoinstitute for Experimental Geochemistry and Geophysics in Germany; NASA’s Jet Propulsion Laboratory; and the University of Chicago.
On the identification of hyperhydrated sodium chloride hydrates, stable at icy moon conditions, Proceedings of the National Academy of Sciences
Astrobiology, Astrochemistry