Potential Cryovolcanic Regions on Ganymede: A Priority Target for JUICE
One of the Jupiter Icy Moons Explorer (JUICE) mission goals of the European Space Agency, launched in 2023 April, is the detailed characterization of Ganymede, Jupiter’s largest moon and a confirmed ocean world.
Among its science objectives, JUICE will investigate Ganymede’s past or possibly ongoing cryovolcanic and tectonic activity and assess material exchange between the surface, subsurface, and internal ocean.
The JUICE Science Working Team has identified potential cryovolcanic regions as targets of high scientific interest, very relevant for astrobiological investigations. Using Voyager and Galileo images, G. C. Collins et al. identified 20 paterae and other candidate cryovolcanic regions, yet their origin and composition remain poorly constrained due to the limited spatial and spectral resolution of existing datasets.
Here, we present a comprehensive reassessment of these 20 regions to support JUICE mission preparation. We integrate detailed analysis with a systematic spectral investigation using reprocessed Galileo Near-Infrared Mapping Spectrometer data and a consistent linear spectral unmixing approach which enables comparative assessment of compositional trends and spectral variability.
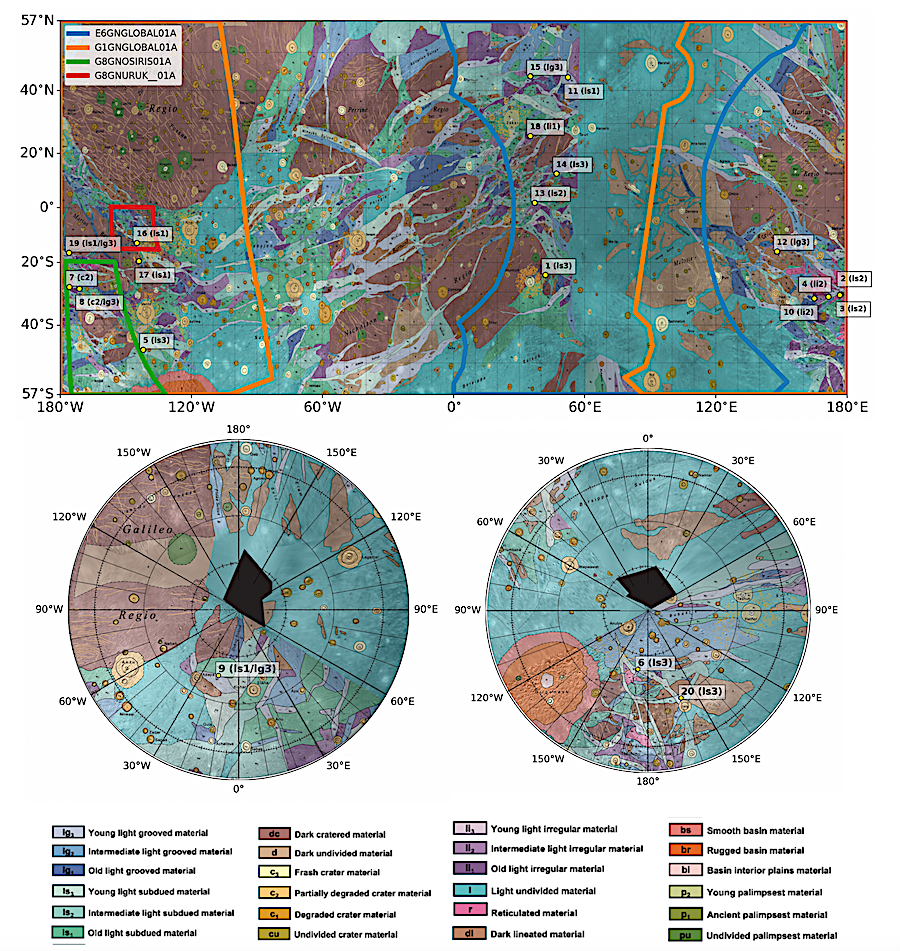
Locations (yellow circles) of ROIs that include paterae, potential flows, and cryovolcanic candidate regions (K. Stephan et al. 2021a) on top of the global geological map of Ganymede from G. C. Collins et al. (2013). (Top: Mercator projection; bottom left: orthogonal projection centered at the north pole; and bottom right: orthogonal projection centered at the south pole.) Each ROI label includes the geologic unit label as described in G. C. Collins et al. (2013). Examples include: light subdued (“ls”) materials; light grooved (“lg”) materials; light irregular (“li”) materials; and crater (“c”) materials. Colored outlines in the Mercator projection represent the areas covered by the four Near-Infrared Mapping Spectrometer (NIMS) cubes included in this study. — The Planetary Science Journal
Our results reveal distinct spectral groups among the candidate paterae, ranging from ice-dominated terrains to salt-enriched assemblages, showing systematic differences across model runs that use temperature-dependent laboratory end-members. Several regions display coherent morphological characteristics and compositional signatures consistent with cryovolcanic resurfacing or brine-related processes.
These regions emerge as the most compelling targets for high-resolution imaging and spectroscopic observations by JUICE instruments, including with the camera JANUS and with MAJIS. This integrated analysis refines the prioritization of candidate cryovolcanic regions and provides a framework for interpreting future JUICE observations of Ganymede.

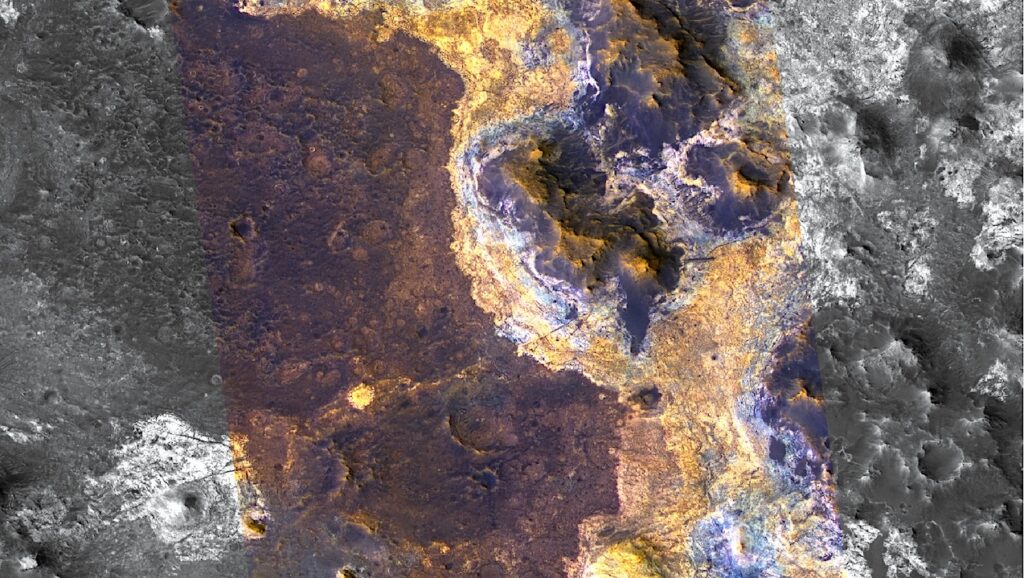
Well-defined potential paterae and flows on Ganymede. The orange arrows correspond to possible caldera-like features and the yellow to possible flow features. Observations by Voyager/NAC1 (1979 March), Voyager/NAC2 (1979 July), and Galileo/SSI (1997 May–June). The coordinates for each ROI, as well as the approximate feature sizes, are listed in Table 1. — The Planetary Science Journal,
Potential Cryovolcanic Regions on Ganymede: A Priority Target for JUICE, The Planetary Science Journal, (open access)
Astrobiology, Astrogeology,