Understanding Aging and Muscle Loss in Space

In space, the human body loses muscle mass. Although living in microgravity requires no heavy lifting, this loss of muscle reduces physical performance.
Decreased muscle mass could also prove particularly problematic on future missions to destinations such as the Moon or Mars. An investigation on the International Space Station aims to understand how this loss occurs so scientists will know more about how to keep astronauts strong.
The Molecular Muscle investigation examines the mechanisms behind muscle loss at the molecular level, and the potential for developing countermeasures targeting those mechanisms. It looks specifically at the activity of genes involved in insulin signaling and cell attachment. Previous research shows that spaceflight affects this activity, leading to muscular and metabolic abnormalities in a variety of organisms.
“Insulin and cell attachment signaling are two of the most consistently changed molecular pathways during spaceflight,” said Timothy Etheridge, a researcher at University of Exeter in the United Kingdom, and one of the principal investigators. Establishing that these changes cause muscle loss during spaceflight makes them a likely target for prevention and treatment measures on future spaceflight missions.
Researchers are using a model organism, the roundworm C. elegans.
“At the molecular level, both structurally and metabolically, the worms are highly similar to humans and show the same changes to spaceflight,” Etheridge said. “In practical terms, they are very small, inexpensive and grow quickly, which reduces the cost and time required for experiments.” C. elegans also has a well-described nervous system of only 302 neurons and its entire genome has been sequenced.
In order to determine whether insulin and cell attachment signaling mechanisms are the underlying cause of muscle loss, the research plans to examine alterations in gene and protein expression and the role of multiple molecular and metabolic pathways affected by spaceflight. In addition, a team of scientists at the Foundation for Research and Technology-Hellas (FORTH) and the University of Crete in Greece plan to examine spaceflight’s role in damage to specific neurons.
Further analysis assesses whether interventions currently used to maintain muscle health actually prevent the gene and protein expression changes repeatedly observed in spaceflight.
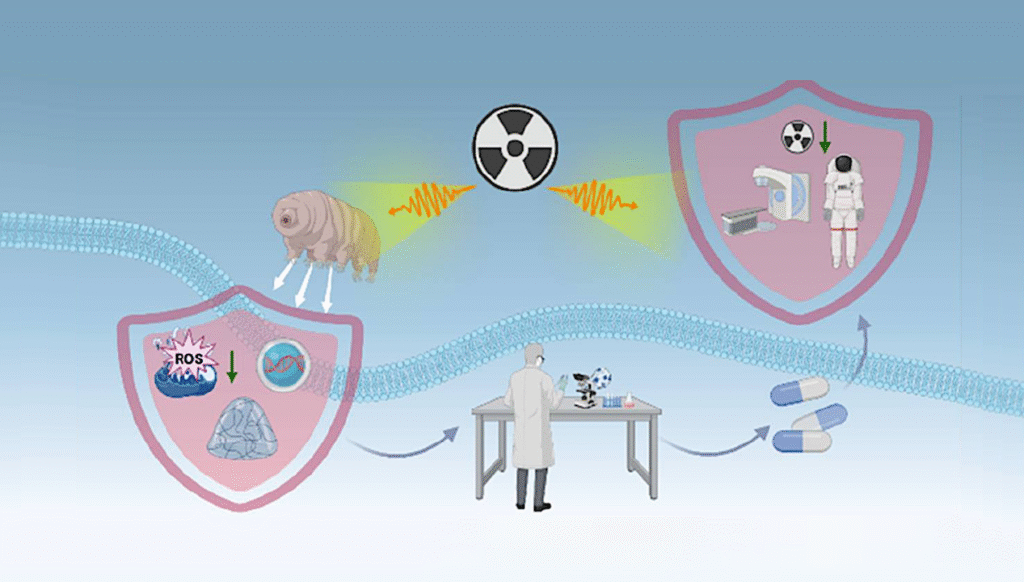
The results may have a number of applications on Earth as well. Insulin and cell attachment signaling play a role in health issues such as diabetes and muscular dystrophy and are associated with the aging process. In addition, people on Earth also experience loss of muscle mass and performance, particularly as a part of aging.
“Identifying the molecular mechanisms behind neuron and muscle deterioration in space also can be effectively used to develop interventions to battle age-associated neurodegenerative conditions on Earth,” said FORTH’s Nektarios Tavernarakis, also a principal investigator on the project. Spaceflight essentially represents an accelerated model of human aging, and an opportunity to better understand aging on Earth.
Molecular Muscle is the first investigation on the space station to be led by a team from the UK. The UK Space Agency joined the space station in 2012, and is a member nation in the European Space Agency (ESA) exploration program, which contributes to space station costs. The investigation is supported by the ESA, UK Space Agency, UK Biotechnology and Biological Sciences Research Council (BBSRC), Medical Research Council (MRC), and Arthritis Research UK. In addition to those from the UK and Greece, researchers from the United States, Japan and South Korea also are participating.
“We plan future experiments targeting more molecular mechanisms in space using a range of drugs and genetic interventions.” Etheridge said. Knowledge gained can support future deep-space missions and help solve aging and other health challenges on Earth.
Astrobiology