Ancient Nitrogenases Are ATP Dependent
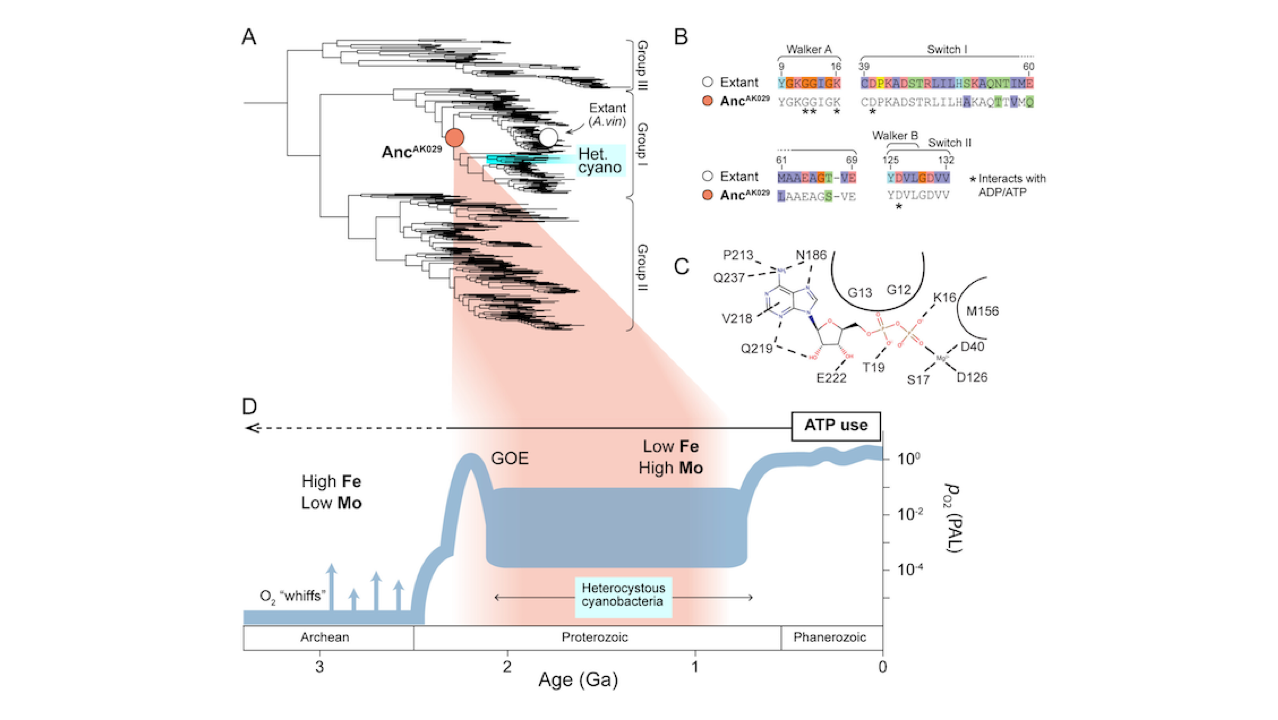
Life depends on a conserved set of chemical energy currencies that are relics of early biochemistry. One of these is ATP, a molecule that, when paired with a divalent metal ion such as Mg2+, can be hydrolyzed to support numerous cellular and molecular processes.
Despite its centrality to extant biochemistry, it is unclear whether ATP supported the function of ancient enzymes. We investigate the evolutionary necessity of ATP by experimentally reconstructing an ancestral variant of the N2-reducing enzyme nitrogenase. The Proterozoic ancestor is predicted to be ~540–2,300 million years old, post-dating the Great Oxidation Event. Growth rates under nitrogen-fixing conditions are ~80% of those of wild type in Azotobacter vinelandii.
In the extant enzyme, the hydrolysis of two MgATP is coupled to electron transfer to support substrate reduction. The ancestor has a strict requirement for ATP with no other nucleotide triphosphate analogs (GTP, ITP, and UTP) supporting activity. Alternative divalent metal ions (Fe2+, Co2+, and Mn2+) support activity with ATP but with diminished activities compared to Mg2+, similar to the extant enzyme.
Additionally, it is shown that the ancestor has an identical efficiency in ATP hydrolyzed per electron transferred to the extant of two. Our results provide direct laboratory evidence of ATP usage by an ancient enzyme.
Ancient nitrogenases are ATP dependent, mBio (open access)
Astrobiology