A Low Temperature Kinetic Study of the C(3P) + CH3OCH3 Reaction. Rate constants, H-atom Product Yields and Astrochemical Implications
Atomic carbon in its ground electronic state, C(3P), is expected to be present at high abundances during the evolution of dense molecular clouds. Consequently, its reactions with other interstellar species could have a strong influence on the chemical composition of these regions.
Here, we report the results of an investigation of the reaction between C(3P) and dimethylether, CH3OCH3, which was recently detected in dark cloud TMC-1. Experiments were performed to study the kinetics of this reaction using a continuous supersonic flow reactor employing pulsed laser photolysis and pulsed laser induced fluorescence for atomic radical generation and detection respectively.
Rate constants for this process were measured between 50 K and 296 K, while additional measurements of the product atomic hydrogen yields were also performed over the 75-296 K range. To better understand the experimental results, statistical rate theory was used to calculate rate constants over the same temperature range and to provide insight on the major product channels.
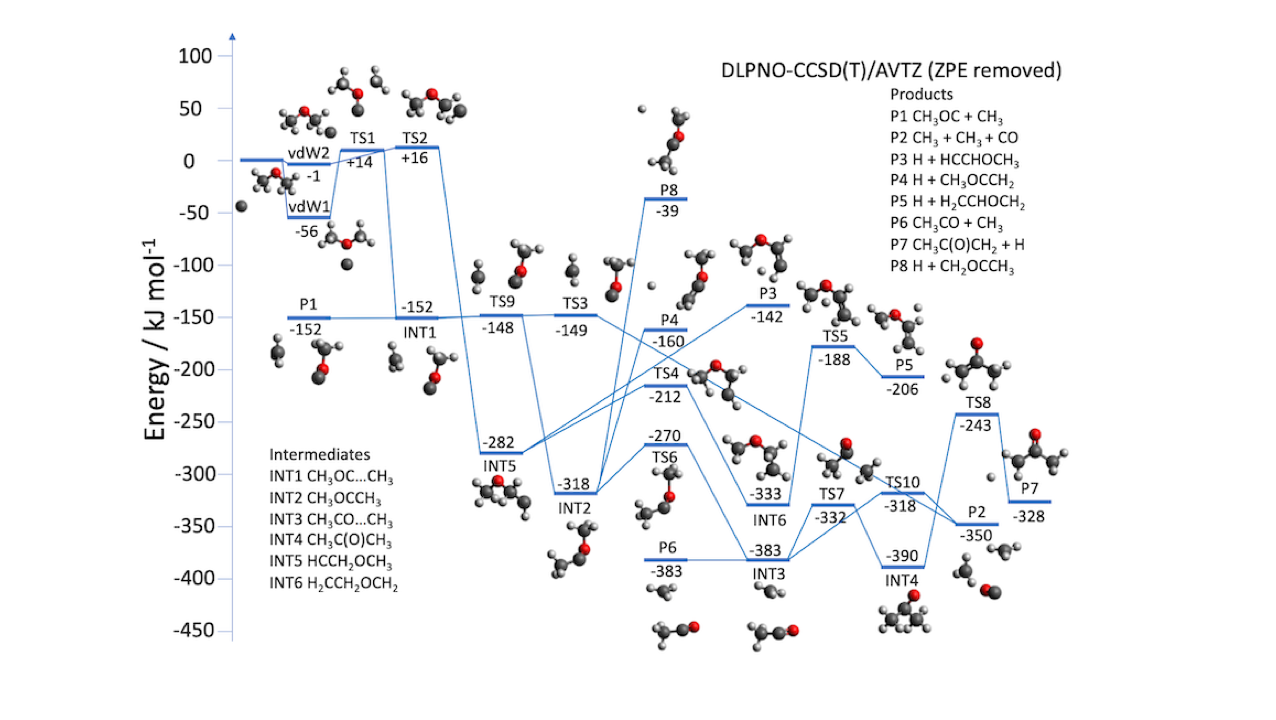
These simulations, based on quantum chemical calculations of the ground triplet state of the C3H6O molecule, allowed us to obtain the most important features of the underlying potential energy surface. The measured rate constant increases as the temperature falls, reaching a value of k_(C+CH_3 OCH_3 )= 7.5 x 10-11 cm3 s-1 at 50 K, while the low measured H-atom yields support the theoretical prediction that the major reaction products are CH3 + CH3 + CO.
The effects of this reaction on the abundances of interstellar CH3OCH3 and related species were tested using a gas-grain dense cloud model, employing an expression for the rate constant, k(T) = alpha(T/300)^beta, with alpha = 1.27 x 10-11 and beta = -1.01. These simulations predict that the C(3P) + CH3OCH3 reaction decreases gas-phase CH3OCH3 abundances by more than an order of magnitude at early times.
Kevin M. Hickson, Jean-Christophe Loison, Valentine Wakelam
Comments: accepted for publication in ACS Earth and Space Chemistry
Subjects: Astrophysics of Galaxies (astro-ph.GA)
Cite as: arXiv:2405.01068 [astro-ph.GA] (or arXiv:2405.01068v1 [astro-ph.GA] for this version)
Related DOI:
https://doi.org/10.1021/acsearthspacechem.4c00014
Focus to learn more
Submission history
From: Kevin Hickson
[v1] Thu, 2 May 2024 07:54:21 UTC (1,716 KB)
https://arxiv.org/abs/2405.01068
Astrobiology, Astrochemistry,