Astrobiology Spinoff? Introduced Tardigrade Proteins Can Slow Metabolism In Human Cells
Editor’s note: although NASA did not sponsor research wherein tardigrade proteins were tested in human cells to see if they work – someone did. This emerged as the result of prior work. Tardigrades are a favorite extremeohpile amongst astrobiologists. They have even been studied on the International Space Station. Drug companies scour the world – sifting through soils, undersea creatures, and toxic waste dumps for interesting organisms that can provide new industrial processes or biomedical applications. One would hope that the people at NASA who do the whole Spinoffs thing every year would pay attention to things like this. Perhaps a little funding might be worth considering. NASA loves to talk about all the things ISS biomedical research can do for people back on Earth. Well, NASA Astrobiology research might also have similar spinoff benefits. NASA won’t know unless it starts to poke around.
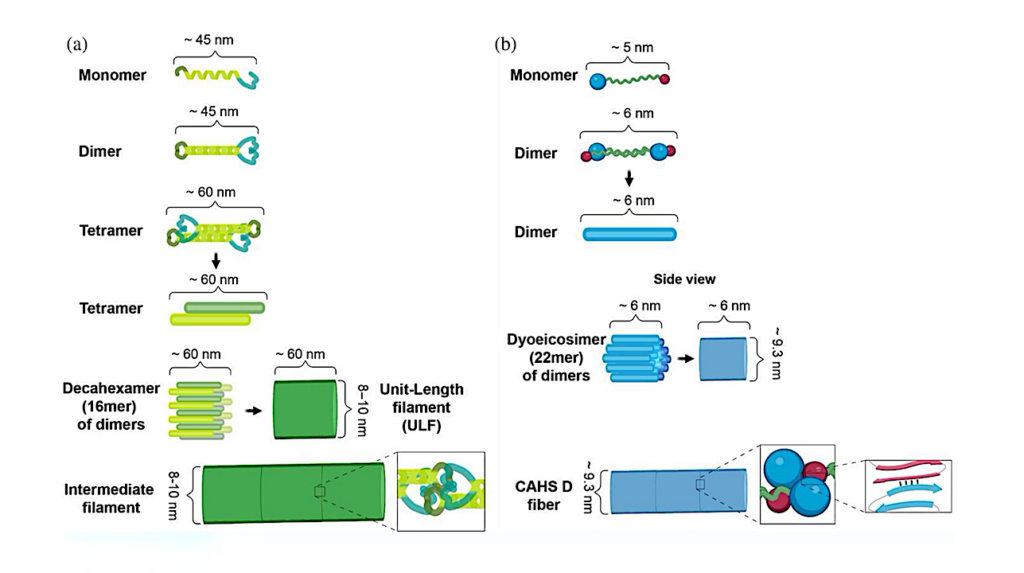
Comparisons of proposed mechanisms of intermediate filament and CAHS D fiber formation. (a) Schematic representation of intermediate filament formation (adapted from (Qin et al., 2009)). (b) Schematic representation of proposed mechanism of CAHS D fiber formation.
University of Wyoming researchers have gained further insight into how tardigrades survive extreme conditions and shown that proteins from the microscopic creatures expressed in human cells can slow down molecular processes.
This makes the tardigrade proteins potential candidates in technologies centered on slowing the aging process and in long-term storage of human cells.
The new study, published in the journal Protein Science, examines the mechanisms used by tardigrades to enter and exit from suspended animation when faced by environmental stress. Led by Senior Research Scientist Silvia Sanchez-Martinez in the lab of UW Department of Molecular Biology Assistant Professor Thomas Boothby, the research provides additional evidence that tardigrade proteins eventually could be used to make life-saving treatments available to people where refrigeration is not possible — and enhance storage of cell-based therapies, such as stem cells.
Measuring less than half a millimeter long, tardigrades — also known as water bears — can survive being completely dried out; being frozen to just above absolute zero (about minus 458 degrees Fahrenheit, when all molecular motion stops); heated to more than 300 degrees Fahrenheit; irradiated several thousand times beyond what a human could withstand; and even survive the vacuum of outer space.
They survive by entering a state of suspended animation called biostasis, using proteins that form gels inside of cells and slow down life processes, according to the new UW-led research. Co-authors of the study are from institutions including the University of Bristol in the United Kingdom, Washington University in St. Louis, the University of California-Merced, the University of Bologna in Italy and the University of Amsterdam in the Netherlands.
Sanchez-Martinez, who came from the Howard Hughes Medical Institute to join Boothby’s UW lab, was the lead author of the paper.
“Amazingly, when we introduce these proteins into human cells, they gel and slow down metabolism, just like in tardigrades,” Sanchez-Martinez says. “Furthermore, just like tardigrades, when you put human cells that have these proteins into biostasis, they become more resistant to stresses, conferring some of the tardigrades’ abilities to the human cells.”
Importantly, the research shows that the whole process is reversible: “When the stress is relieved, the tardigrade gels dissolve, and the human cells return to their normal metabolism,” Boothby says.
“Our findings provide an avenue for pursuing technologies centered on the induction of biostasis in cells and even whole organisms to slow aging and enhance storage and stability,” the researchers concluded.
Previous research by Boothby’s team showed that natural and engineered versions of tardigrade proteins can be used to stabilize an important pharmaceutical used to treat people with hemophilia and other conditions without the need for refrigeration.
Tardigrades’ ability to survive being dried out has puzzled scientists, as the creatures do so in a manner that appears to differ from a number of other organisms with the ability to enter suspended animation.
Labile assembly of a tardigrade protein induces biostasis, Protein Science (open access)
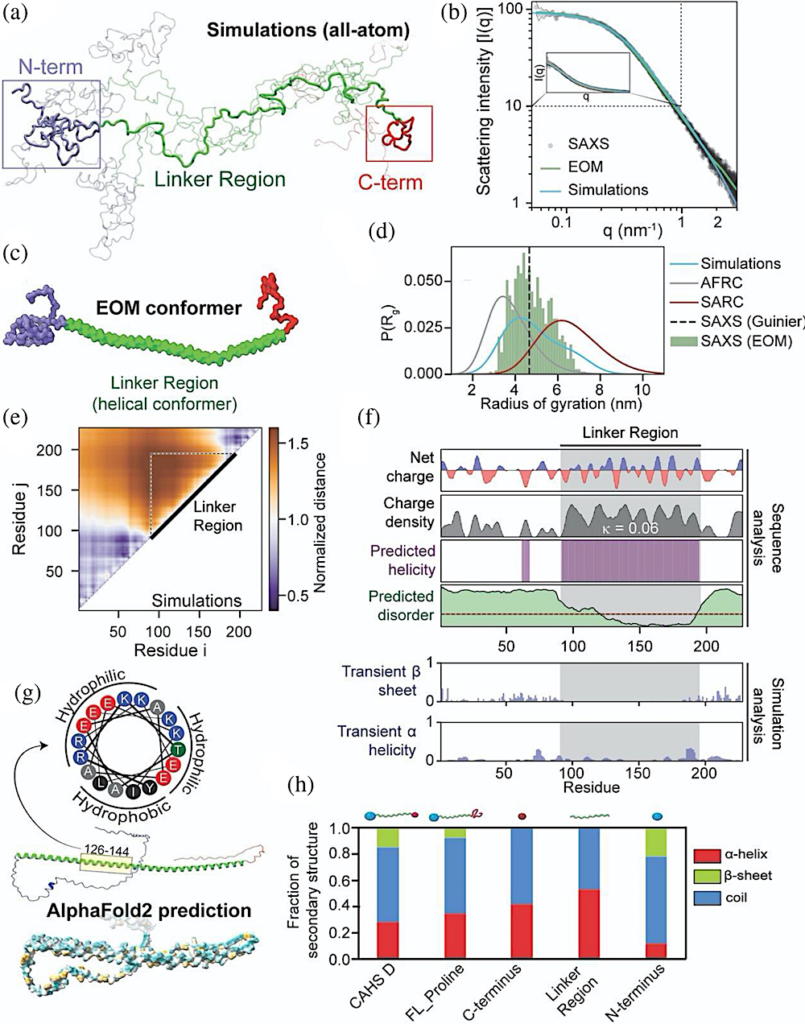
Global and local ensemble features of CAHS D. (a) Representative depiction of the CAHS D ensemble derived from all-atom Monte Carlo simulation. Simulations predict two collapsed termini held apart by an extended linker. (b) SAXS data (black), EOM fitted scattering profile (green), and simulation-derived scattering profile (blue) superimposed with one another. EOM and simulations are in good agreement with experimental data. See also Figure S2B–F. (c) Representative depiction of the CAHS D ensemble derived from EOM analysis. The three domains observed in simulations (a) are highlighted in blue (N-terminus), green (linker), and red (C-terminus). (d) Comparison of the radius of gyration distributions for monomeric CAHS D obtained from SAXS (dotted line), EOM (green bars), and Monte Carlo simulation (blue curve). For comparison, overlays for the Gaussian-chain-like AFRC model (gray) and self-avoiding random coil (red) generated from sequence-matched ensembles are shown. (e) A normalized distance map from CAHS D simulations compares the average inter-residue distance with the expected inter-residue distance if CAHS D behaved like a Gaussian chain. Hotter colors are further apart, cooler colors are closer. This analysis suggests that the terminal regions are largely collapsed, while the linker is expanded. (f) Localization of different sequence features within the CAHS D ensemble using bioinformatics and simulation analysis. (g) (bottom) Alphafold2 predictions of the CAHS D ensemble suggest high amphipathic helical propensity within the linker region (dark cyan most hydrophilic, dark golden most lipophilic). Helical wheel diagram of residues 126–144 of CAHS D (top). (h) Circular dichroism spectroscopy of CAHS D showing the fraction of different types of secondary structure for different variants.
Astrobiology