Unlocking Long-term Genetic Memory: Dormant Bacterial Spores Offer Key Insights Into Evolutionary Survival Strategies

A recent study spearheaded by Prof. Sigal Ben Yehuda and her team at Hebrew University has unveiled a captivating facet of bacterial dormancy. This research illuminates the mechanism through which dormant bacterial spores uphold and activate an enduring transcriptional program upon revival, showcasing an extraordinary genetic memory system.
This discovery is pivotal as it unravels the mechanisms governing how these organisms retain vital genetic information during years of dormancy. Understanding this process not only sheds light on bacterial survival in harsh conditions but also holds broader implications. It could offer insights into sustaining long-term transcriptional programs in diverse organisms, potentially impacting fields such as microbiology, biotechnology, and medicine. Such knowledge might pave the way for strategies to control pathogens, enhance biotechnological processes, and deepen our understanding of dormant states across different life forms.
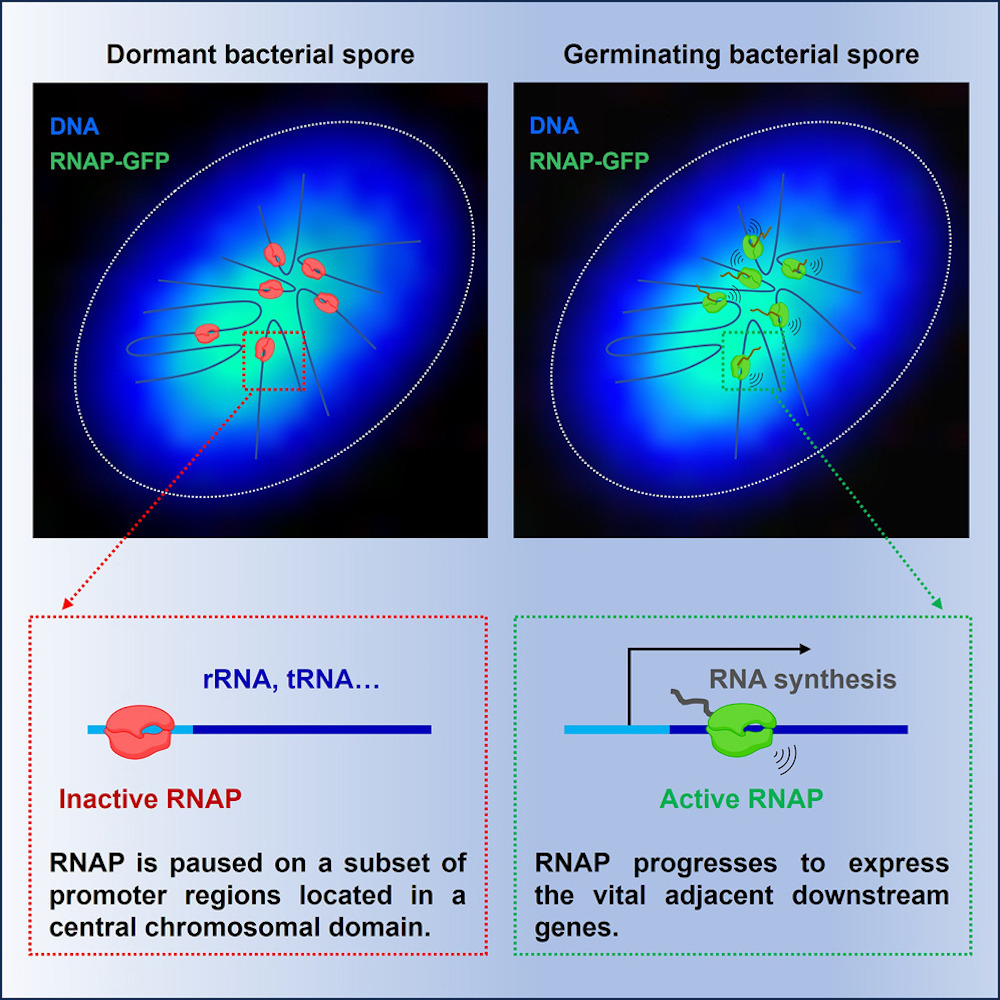
Graphical abstract
Spores, resilient and protective structures formed by certain microorganisms like bacteria and fungi, serve as a survival mechanism against adverse conditions. Bacterial spores are among the longest-living cellular forms on Earth, with reports evoking their revival following millions of years of quiescence. They encapsulate the organism’s genetic material and essential components, remaining dormant until conditions become favorable for germination. Comprehending spores is crucial in various fields, offering insights into survival strategies and potential applications in microbiology, agriculture, and biotechnology.
The study, published in Molecular Cell, highlights the discovery of a central chromosomal domain within dormant spores. This domain hosts core RNA polymerase (RNAP), which remains bound to specific intergenic promoter regions during dormancy. These regions exert control over genes crucial for essential cellular functions, such as the production of rRNAs and tRNAs.
Upon emergence from dormancy, the RNA polymerase inside these spores promptly initiates the copying of vital genetic instructions. It recruits necessary components for transcription, such as sigma factors, swiftly activating essential genes necessary for cellular functions. The study also observed a similar process in disease-causing bacteria that form spores, suggesting a common strategy among various organisms to reinitiate functions after dormancy.
The research also uncovered the pivotal role of spore DNA-compacting proteins in this process. Mutants lacking these proteins displayed scattered RNAP localization, resulting in disorganized gene expression during germination. This underscores the significance of maintaining proper chromosomal structure in preserving the transcriptional program crucial for spore revival.
Prof. Sigal Ben Yehuda remarked, “Our research suggests that the structure of the spore chromosome is designed to uphold a blueprint for gene activity by pausing RNA polymerase, in a standby mode, ready to resume gene expression when conditions favor revival. This mechanism’s relevance might extend beyond bacteria, offering valuable insights into maintaining enduring gene activity plans across various organisms that undergo dormant life stages.”
This research signifies a substantial leap in comprehending the intricate mechanisms behind bacterial dormancy and revival. Its implications span diverse fields, from microbiology to potential applications in biotechnology and medicine.
Dormant bacterial spores encrypt a long-lasting transcriptional program to be executed during revival, Molecular Cell
Astrobiology, Microbiology,