Building Blocks For Life Could Have Formed Near New Stars And Planets
While life on Earth is relatively new, geologically speaking, the ingredients that combined to form it might be much older than once thought.
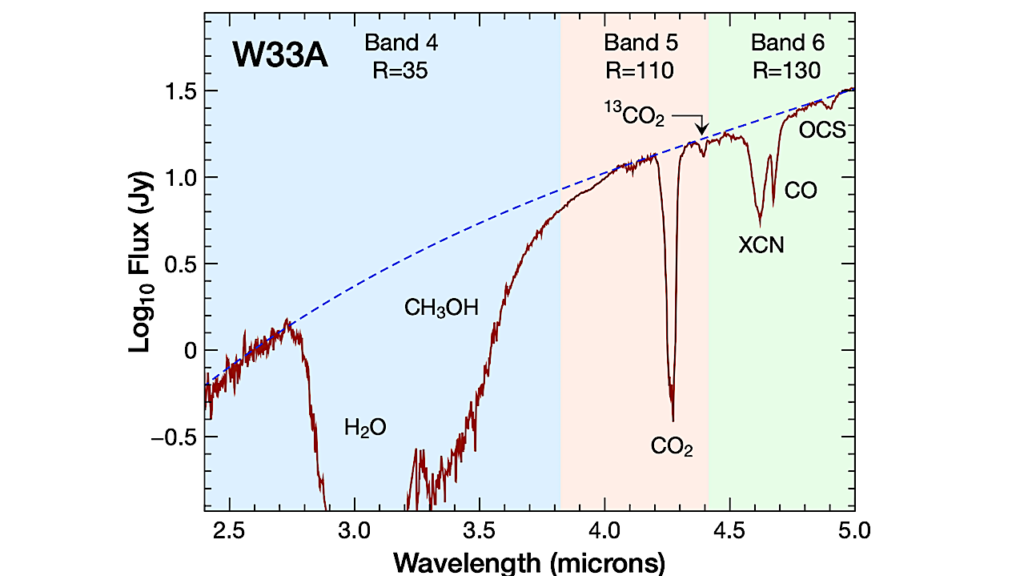
According to research published in ACS Central Science, the simplest amino acid, carbamic acid, could have formed alongside stars or planets within interstellar ices. The findings could be used to train deep space instruments like the James Webb Space Telescope to search for prebiotic molecules in distant, star-forming regions of the universe.
It has long been hypothesized that one of the building blocks for life, amino acids, could have formed during reactions in the “primordial soup” of the early, prebiotic Earth. However, another theory suggests that amino acids could have been carried to the Earth’s surface by meteorites. These space rocks might have picked up the molecules from dust or interstellar ices — water and other gases frozen solid by the cold temperatures of outer space.
But because meteorites came from far away in the universe, scientists are left wondering, where did these molecules form, and when? To help answer these questions, Ralf Kaiser, Agnes Chang and colleagues wanted to investigate the chemical reactions that might have taken place in interstellar ices that once existed near newly forming stars and planets.
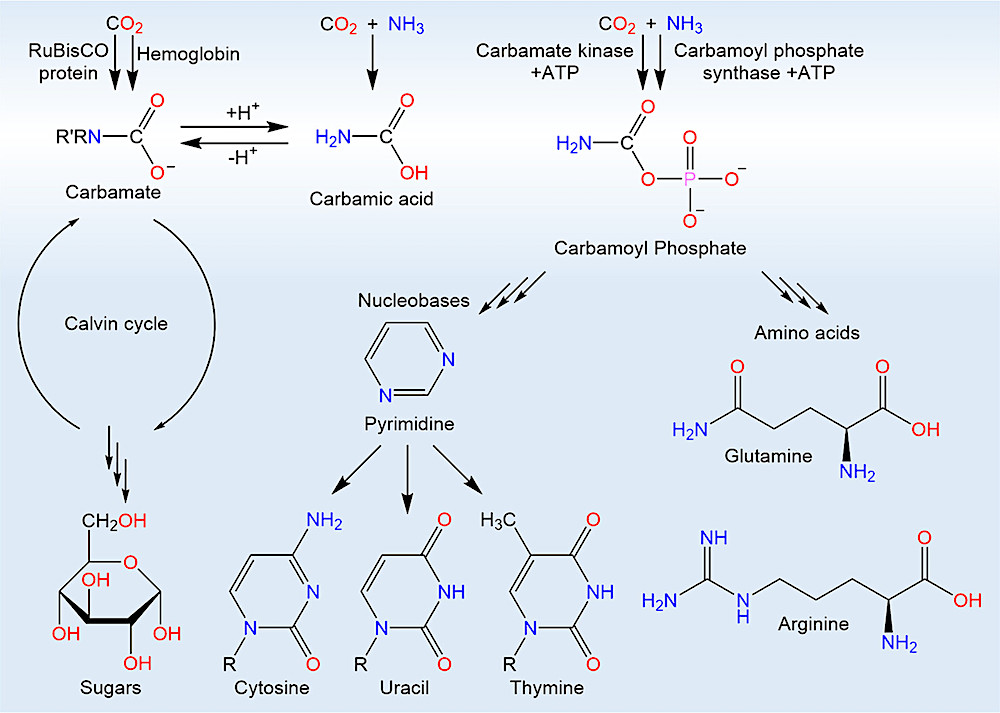
Additions of ammonia and amines to carbon dioxide are fundamental reactions in contemporary biochemical pathways. Biological sugar synthesis is initiated by carbon dioxide capture with amines, as is carbon dioxide transport by hemoglobin. Formation of carbamoyl phosphate is the first step in the synthesis of pyrimidine-derived nucleobases and the amino acids glutamine and arginine.
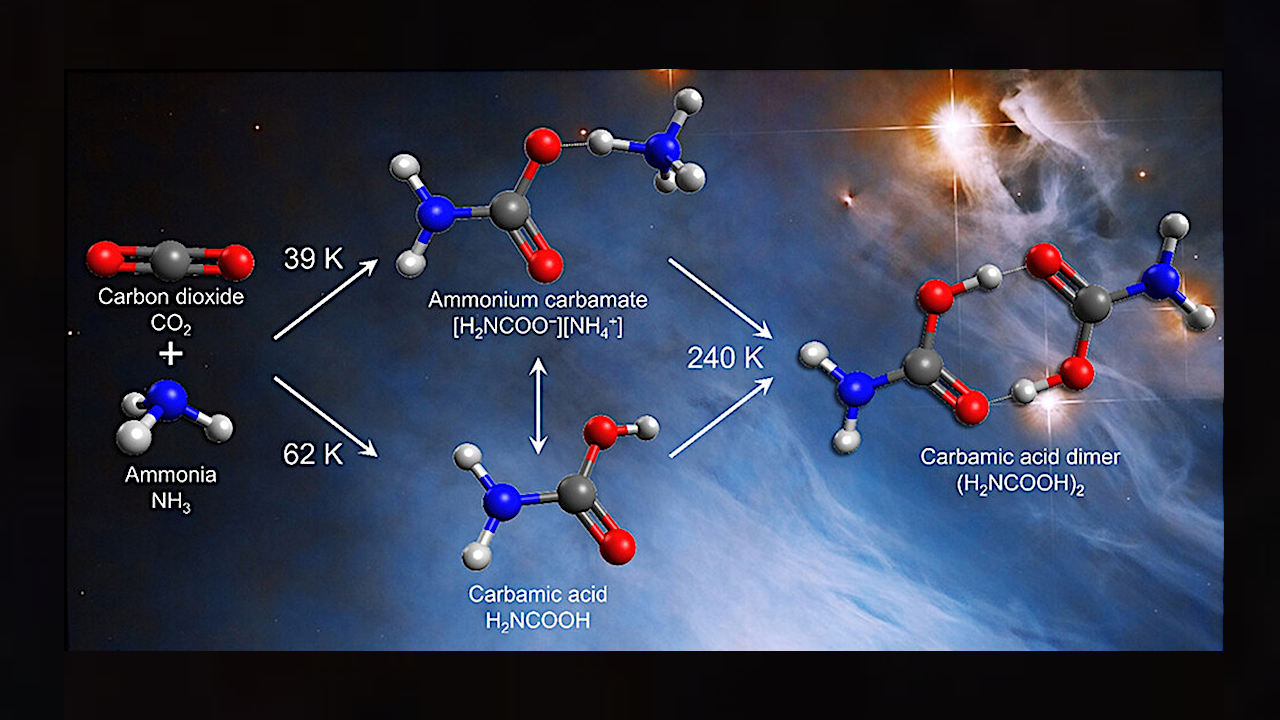
The team created model interstellar ices containing ammonia and carbon dioxide, which were deposited onto a silver substrate and slowly heated. Using Fourier transform infrared spectroscopy, they found that carbamic acid and ammonium carbamate started to form at -348 degrees Fahrenheit and -389 degrees (62 and 39 Kelvin), respectively.
These low temperatures demonstrate that these molecules — which can turn into more complex amino acids — could have formed during the earliest, coldest stages of star formation. In addition, the researchers found that at warmer temperatures, similar to those produced by a newly formed star, two carbamic acid molecules could link together, making a stable gas.
The team hypothesized that these molecules could have been incorporated into the raw materials of solar systems including our own and then delivered to the early Earth by comets or meteorites once the planet formed. They hope this work will inform future studies that use powerful telescopes to look for evidence of prebiotic molecules in the far reaches of space.
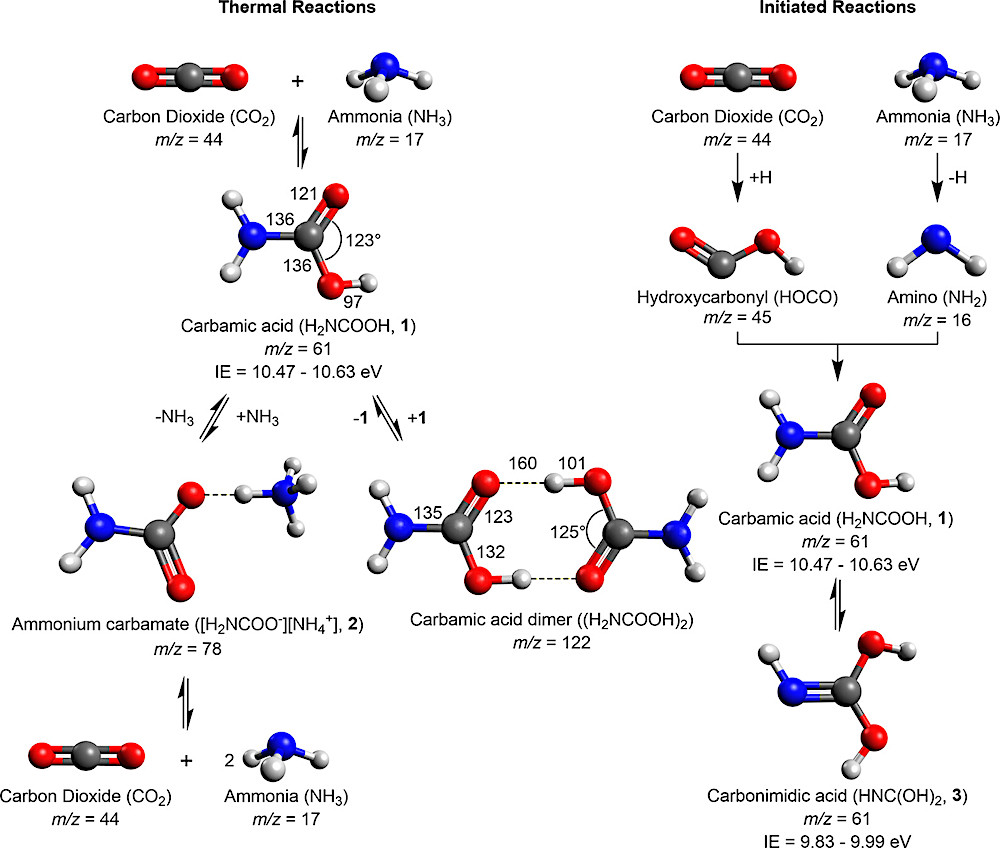
Reaction scheme for thermal and initiated reactions of carbon dioxide and ammonia in interstellar analog ices including mass-to-charge ratio (m/z) and ionization energy (IE). Carbamic acid (H2NCOOH, 1) can be formed by addition of ammonia to carbon dioxide or by radical-radical recombination. Ammonium carbamate ([H2NCOO–][NH4+], 2) can be formed by acid-base reactions of 1 and ammonia or by termolecular reactions of two molecules of ammonia with carbon dioxide. Amide-iminol tautomerization of 1 may result in carbonimidic acid (HNC(OH)2, 3) with initiation. Calculated structural parameters are in units of pm for bond lengths and degrees for bond angles.
The authors acknowledge funding from the Division for Astronomy of the U.S. National Science Foundation, the W.M. Keck Foundation and the University of Hawaii at Manoa.
Thermal Synthesis of Carbamic Acid and Its Dimer in Interstellar Ices: A Reservoir of Interstellar Amino Acids, ACS Cent. Sci. (open access)
Abstract
Reactions in interstellar ices are shown to be capable of producing key prebiotic molecules without energetic radiation that are necessary for the origins of life. When present in interstellar ices, carbamic acid (H2NCOOH) can serve as a condensed-phase source of the molecular building blocks for more complex proteinogenic amino acids. Here, Fourier transform infrared spectroscopy during heating of analogue interstellar ices composed of carbon dioxide and ammonia identifies the lower limit for thermal synthesis to be 62 ± 3 K for carbamic acid and 39 ± 4 K for its salt ammonium carbamate ([H2NCOO–][NH4+]). While solvation increases the rates of formation and decomposition of carbamic acid in ice, the absence of solvent effects after sublimation results in a significant barrier to dissociation and a stable gas-phase molecule. Photoionization reflectron time-of-flight mass spectrometry permits an unprecedented degree of sensitivity toward gaseous carbamic acid and demonstrates sublimation of carbamic acid from decomposition of ammonium carbamate and again at higher temperatures from carbamic acid dimers. Since the dimer is observed at temperatures up to 290 K, similar to the environment of a protoplanetary disk, this dimer is a promising reservoir of amino acids during the formation of stars and planets.
Astrobiology, Astrochemistry,