Fully Fluorinated Non-carbon Compounds NF3 And SF6 As Ideal Technosignature Gases
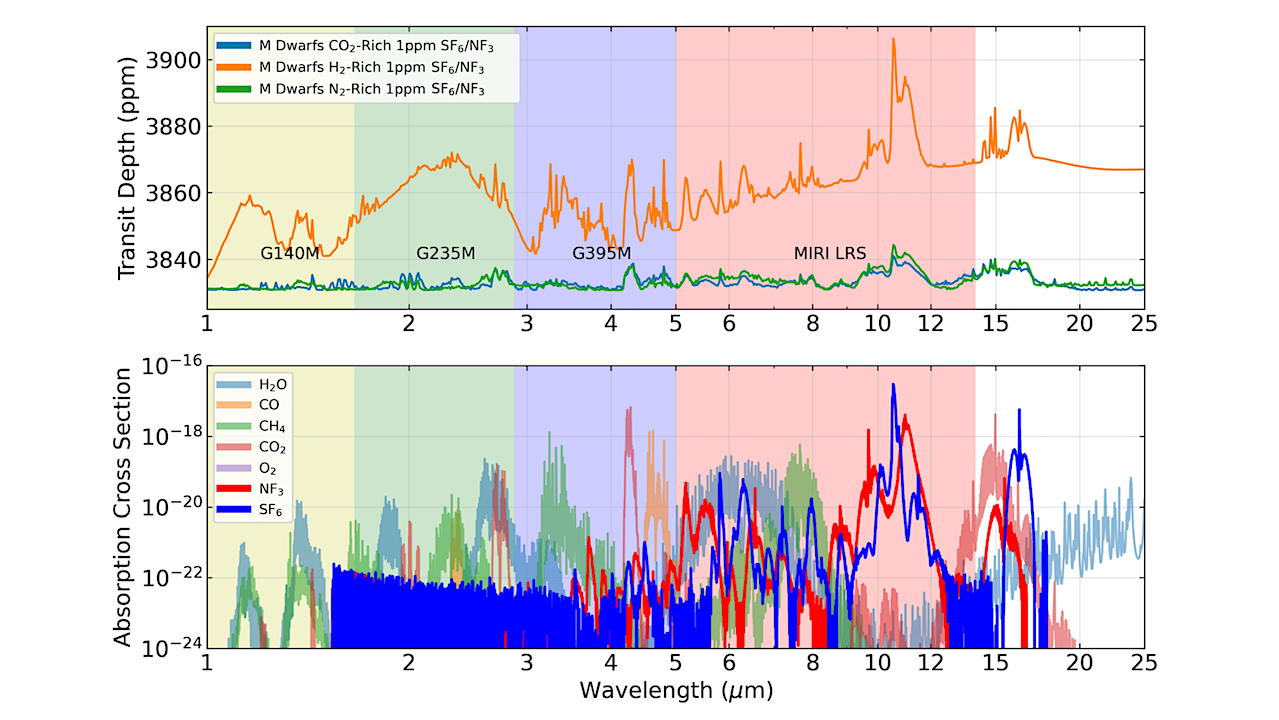
Waste gas products from technological civilizations may accumulate in an exoplanet atmosphere to detectable levels. We propose nitrogen trifluoride (NF3) and sulfur hexafluoride (SF6) as ideal technosignature gases.
Earth life avoids producing or using any N-F or S-F bond-containing molecules and makes no fully fluorinated molecules with any element. NF3 and SF6 may be universal technosignatures owing to their special industrial properties, which unlike biosignature gases, are not species-dependent. Other key relevant qualities of NF3 and SF6 are: their extremely low water solubility, unique spectral features, and long atmospheric lifetimes.
NF3 has no non-human sources and was absent from Earth’s pre-industrial atmosphere. SF6 is released in only tiny amounts from fluorine-containing minerals, and is likely produced in only trivial amounts by volcanic eruptions. We propose a strategy to rule out SF6’s abiotic source by simultaneous observations of SiF4, which is released by volcanoes in an order of magnitude higher abundance than SF6.
Other fully fluorinated human-made molecules are of interest, but their chemical and spectral properties are unavailable. We summarize why life on Earth-and perhaps life elsewhere-avoids using F. We caution, however, that we cannot definitively disentangle an alien biochemistry byproduct from a technosignature gas.
Sara Seager, Janusz J. Petkowski, Jingcheng Huang, Zhuchang Zhan, Sai Ravela, William Bains
Comments: Published as a part of the special collection “Exoplanets” in Scientific Reports
Subjects: Earth and Planetary Astrophysics (astro-ph.EP)
Cite as: arXiv:2308.13667 [astro-ph.EP] (or arXiv:2308.13667v1 [astro-ph.EP] for this version)
Journal reference: Scientific Reports 13.1 (2023): 13576
Related DOI:
https://doi.org/10.1038/s41598-023-39972-z
Focus to learn more
Submission history
From: Janusz Petkowski
[v1] Fri, 25 Aug 2023 20:48:15 UTC (3,317 KB)
https://arxiv.org/abs/2308.13667
Astrobiology, Astrochemistry,