Cracking the Puzzle of CO2 Formation on Interstellar Ices. Quantum Chemical and Kinetic Study of the CO + OH -> CO2 + H Reaction
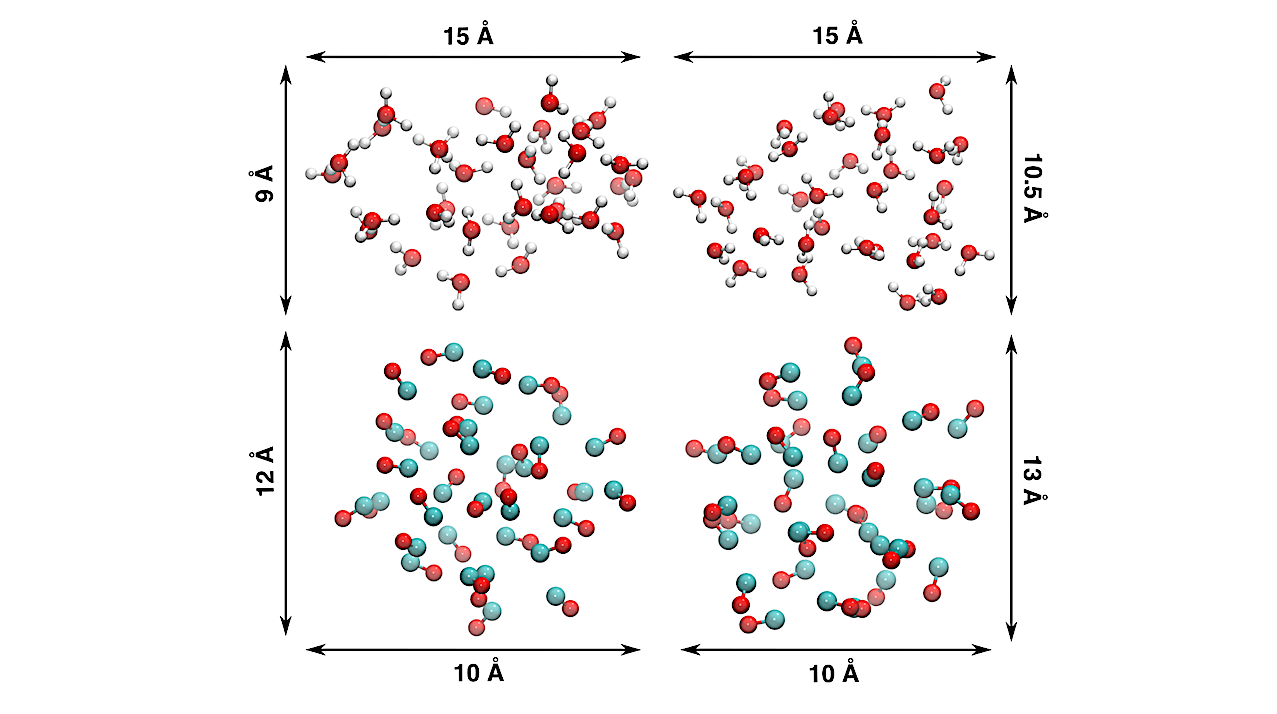
CO2 is one of the dominant components of the interstellar ice. Recent observations show CO2 exists more abundantly in polar (H2O-dominated) ice than in apolar (H2O-poor) ice. CO2 ice formation is primarily attributed to the reaction between CO and OH, which has a barrier.
Highly accurate quantum chemical calculations were employed to analyze the stationary points of the potential energy surfaces of the title reaction in the gas phase on a H2O and CO clusters. Microcanonical transition state theory was used as a diagnostic tool for the efficiency of the reaction under ISM conditions.
We simulate the kinetics of ice chemistry, considering different scenarios involving non-thermal processes and energy dissipation. The CO + OH reaction proceeds through the remarkably stable intermediate HOCO radical. On the H2O cluster, the formation of this intermediate is efficient, but the subsequent reaction leading to CO2 formation is not. Conversely, HOCO formation on the CO cluster is inefficient without external energy input.
Thus, CO2 ice cannot be formed by the title reaction alone either on the H2O cluster or CO cluster. In the polar ice, CO2 ice formation is possible via CO + OH -> HOCO, followed by HOCO + H ->CO2 + H2, as demonstrated by abundant experimental literature. In apolar ice, CO2 formation is less efficient because HOCO formation requires external energy. Our finding is consistent with the JWST observations. Further experimental work is encouraged using low-temperature OH radicals.
Germán Molpeceres, Joan Enrique-Romero, Yuri Aikawa
Comments: Accepted for publication in Astronomy and Astrophysics
Subjects: Astrophysics of Galaxies (astro-ph.GA)
Cite as: arXiv:2307.04088 [astro-ph.GA] (or arXiv:2307.04088v1 [astro-ph.GA] for this version)
https://doi.org/10.48550/arXiv.2307.04088
Focus to learn more
Submission history
From: German Molpeceres
[v1] Sun, 9 Jul 2023 03:44:48 UTC (9,064 KB)
https://arxiv.org/abs/2307.04088
Astrobiology, Astrochemistry