Disequilibria From Dead To Living Worlds

Chemical disequilibrium in exoplanetary atmospheres (detectable with remote spectroscopy) can indicate life.
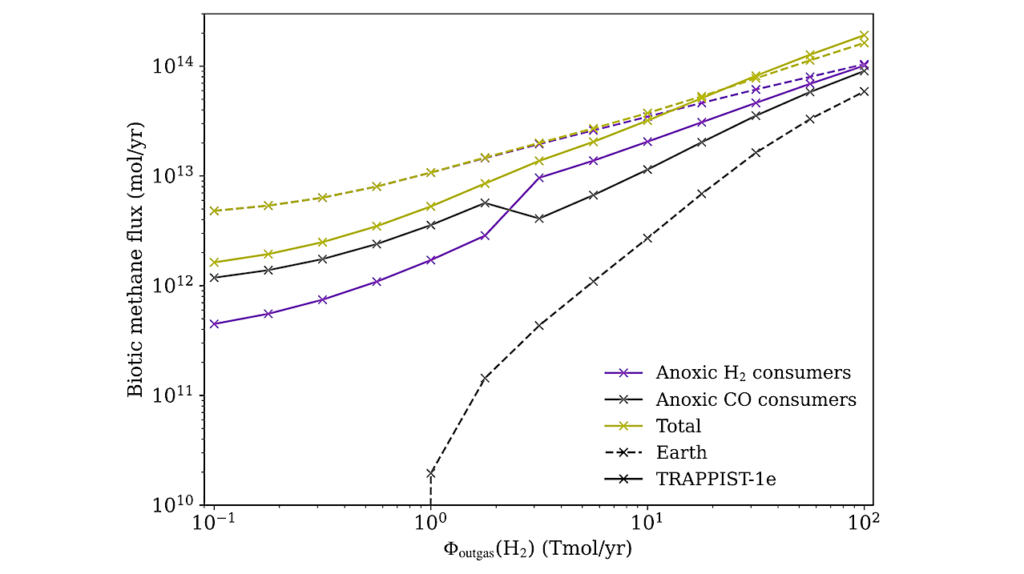
The modern Earth’s atmosphere-ocean system has a much larger chemical disequilibrium than other solar system planets with atmospheres because of oxygenic photosynthesis. However, no analysis exists comparing disequilibrium on lifeless, prebiotic planets to disequilibrium on worlds with primitive chemotrophic biospheres that live off chemicals and not light. Here, we use a photochemical-microbial ecosystem model to calculate the atmosphere-ocean disequilibria of Earth with no life and with a chemotrophic biosphere.
We show that the prebiotic Earth likely had a relatively large atmosphere-ocean disequilibrium due to the coexistence of water and volcanic H2, CO2, and CO. Subsequent chemotrophic life likely destroyed nearly all of the prebiotic disequilibrium through its metabolism, leaving a likely smaller disequilibrium between N2, CO2, CH4, and liquid water. So, disequilibrium fell with the rise of chemotrophic life then later rose with atmospheric oxygenation due to oxygenic photosynthesis.
We conclude that big prebiotic disequilibrium between H2 and CO2 or CO and water is an anti-biosignature because these easily metabolized species can be eaten due to redox reactions with low activation energy barriers. However, large chemical disequilibrium can also be a biosignature when the disequilibrium arises from a chemical mixture with biologically insurmountable activation energy barriers, and clearly identifiable biogenic gases. The modern disequilibrium between O2, N2, and liquid water along with minor CH4 is such a case. Thus, the interpretation of disequilibrium requires context. With context, disequilibrium can be used to infer dead or living worlds.
When is chemical disequilibrium in Earth-like planetary atmospheres a biosignature versus an anti-biosignature? Disequilibria from dead to living worlds
Nicholas Wogan, David Catling
(Submitted on 15 Nov 2019)
Comments: Submitted this draft to Astrophysical Journal on 11/14/19
Subjects: Earth and Planetary Astrophysics (astro-ph.EP)
Cite as: arXiv:1911.06852 [astro-ph.EP] (or arXiv:1911.06852v1 [astro-ph.EP] for this version)
Submission history
From: Nicholas Wogan
[v1] Fri, 15 Nov 2019 19:48:29 UTC (2,133 KB)
https://arxiv.org/abs/1911.06852
Astrobiology